34 vapour liquid equilibrium diagram
It is assumed that the mixture is maintained at equilibrium, thus the pressure above the liquid is equal to the saturation pressure of the mixture. For ideal mixtures (hydrocarbon mixtures typically) it is possible to calculate the vapor pressure thanks to the composition of the liquid phase, this equation is called Raoult's law. 2 Phase diagrams combine these behaviors in a T,P plot showing regions of stability Consider processes: a, green region solid, const. P, increase T, to 1st black line solid-liquid equilibrium, then blue area liquid, to 2nd black line liquid-vapor equilibrium, beige region - gas b, start solid (green) to black line solid-vapor equilibrium, then vapor (beige)
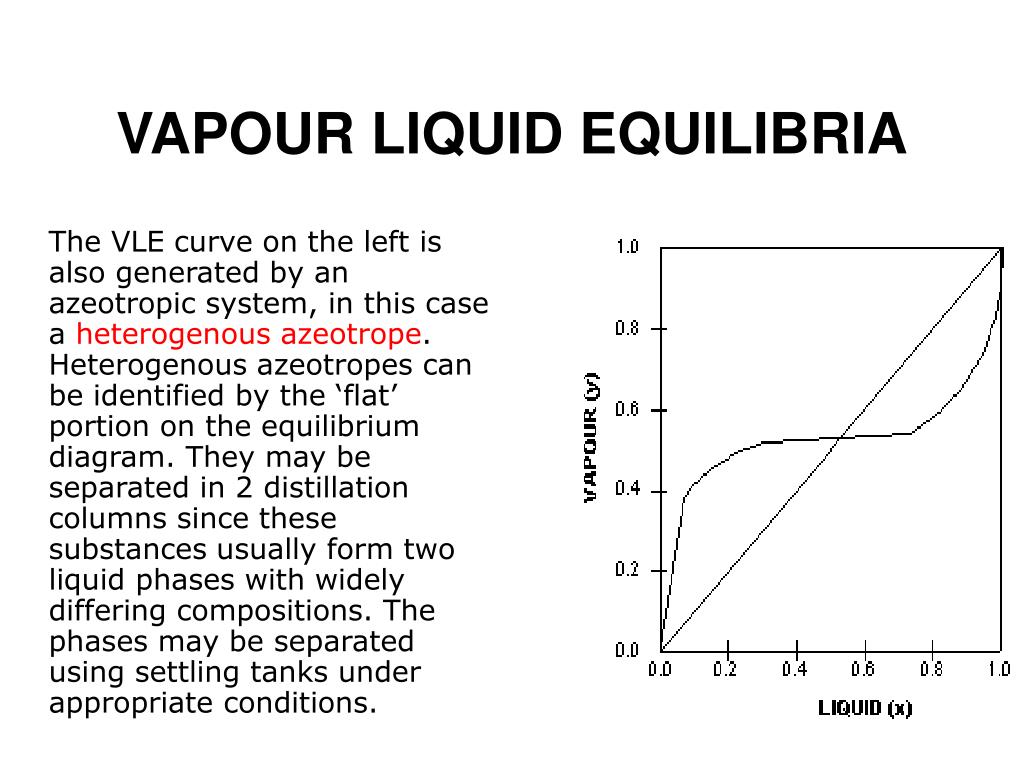
Thus the sizes, particularly the height, of distillation columns are determined by the vapour liquid equilibrium (VLE) data for the mixtures. Vapour-Liquid-Equilibrium (VLE) Curves. Constant pressure VLE data is obtained from boiling point diagrams. VLE data of binary mixtures is often presented as a plot, as shown in the figure on the right.
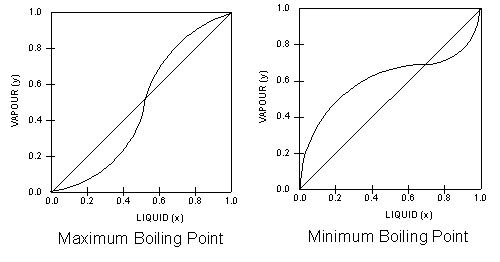
Vapour liquid equilibrium diagram
Uses an interactive simulation to explain how to read a T-x-y diagram for a binary mixture that obeys Raoult's law. This simulation is located at: https://w... Txy Diagram. Txy diagram plots bubble and dew point curves at constant pressure P. Put down the liquid mol fraction x 1 from 0.0 to 1.0 with increment of 0.01 in spreadsheet. Iteration is done for each liquid mol fraction to estimate equilibrium temperature T and activity coefficient γ i. In thermodynamics and chemical engineering, the vapor–liquid equilibrium (VLE) describes the distribution of a chemical species between the vapor phase and a liquid phase.. The concentration of a vapor in contact with its liquid, especially at equilibrium, is often expressed in terms of vapor pressure, which will be a partial pressure (a part of the total gas pressure) if any other gas(es ...
Vapour liquid equilibrium diagram. SIMPLE MODELS FOR VAPOR/LIQUID EQUILIBRIUM 3.1 Raoult's Law • Assumptions: - The vapor phase is an ideal gas (low to moderate pressure) - The liquid phase is an ideal solution (the system are chemically similar) *Chemically similar: the molecular species are not too different in size and are of the same chemical nature. eg: n-hexane/n ... Draw vapor-liquid equilibrium (VLE) and liquid-miscibility (LLE) phase diagrams, or calculate distillation processes. Over 300 organic compounds. This Demonstration shows and diagrams for vapor-liquid equilibrium (VLE) of a benzene/ethanol mixture. This liquid mixture is non-ideal, and the system has an azeotrope (a condition where liquid and vapor have the same composition). The blue curve represents the liquid-phase boundary (bubble point), and the green curve represents the vapor ... Vapour/liquid equilibrium is the state of coexistence of liquid and vapour phases. In the qualitative discussion, we shall focus on systems comprised of two chemical species, because systems of greater complexity cannot be adequately represented graphically. When N = 2 the phase rule becomes F = 4 ˇ. Since there must be at least one
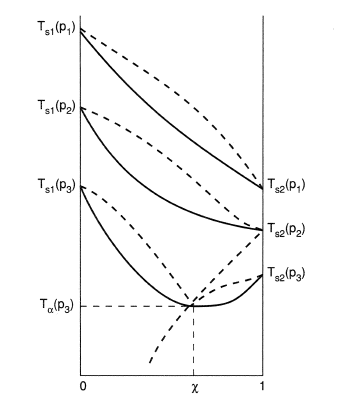
A schematic three-dimensional diagram illustrating these surfaces for VLE is shown in Fig. 10.1. This figure shows schematically the P-T-composition surfaces which contain the equilibrium states of saturated vapor and saturated liquid for a binary system. 7.2 Three dimensional VLE phase diagram for a binary system. Consider first a case of VLE phase behaviour at a constant temperature. This is in shown as the.39 pages In relation to the melting point, the triple point of a material occurs when the vapor, liquid, and solid phases are all in equilibrium (Table 4.10). This is the point on a phase diagram where the solid-vapor, solid-liquid, and liquid-vapor equilibrium lines all meet. 19 Apr 2018 — This Demonstration leads you through the construction of a temperaturecomposition diagram step by step for vaporliquid equilibrium VLE of an ...
Presents solution in which 20% of a liquid is vaporized, the vapor is condensed, and 20% of the new liquid is vaporized, and then condensed. The composition ... The basis of distillation is phase equilibrium—specifically, vapor-liquid equilibrium (VLE) and in some cases vapor-liquid-liquid equilibrium (VLLE). Distillation can only ... represents the vapor composition. The second diagram is a plot of x versus y. These types of diagrams are generated at a constant pressure. Because the pressure in a Vapor/Liquid Equilibrium • Previous chapters dealt with pure substances or with constant composition mixtures (air). • But in chemical reactions and number of industrially mass-transfer operations the . composition changes are the desired outcome. (Process such as distillation, absorption and extraction bring phases of different Chapter 8 Phase Diagrams. (b) The interpretation of diagrams. Point a represents the vapor pressure of a mixture with liquid composition xA and b represents the composition of the vapor that is in equilibrium with the liquid at that pressure. Note that when two phases are in equilibrium, P = 2, so F' = 1.
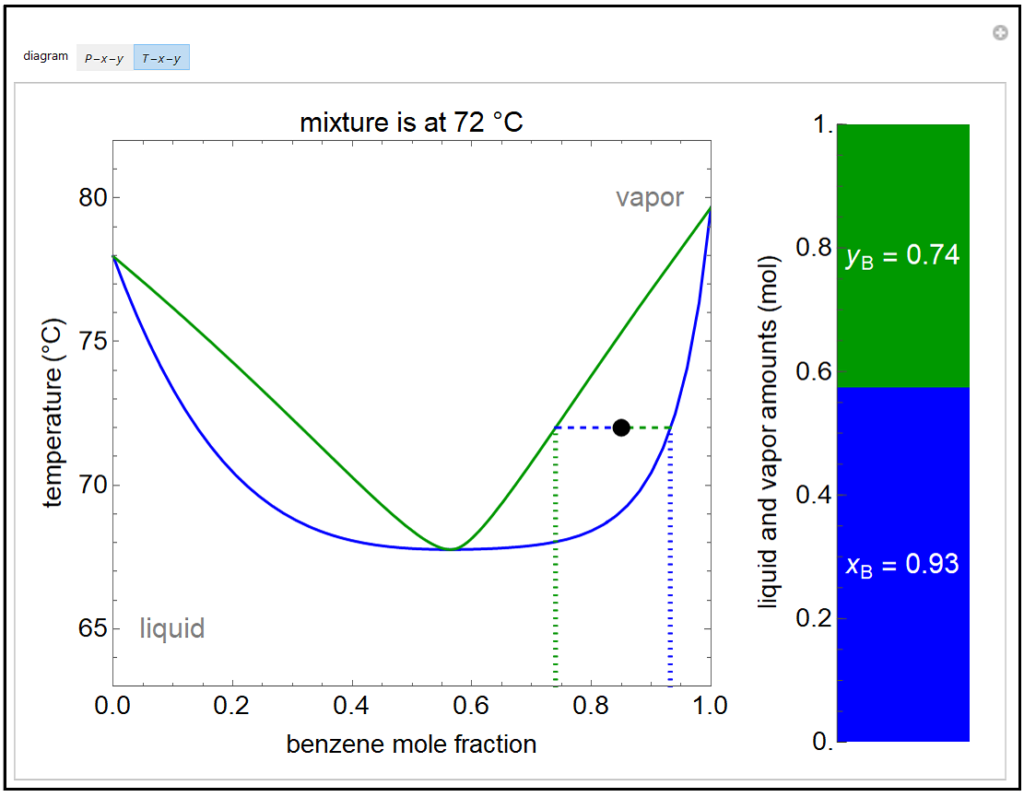
The vapor-liquid equilibrium (VLE) behavior of an -hexane/ -octane mixture is demonstrated in and diagrams. The blue line represents the liquid-phase boundary (bubble point) and the green line represents the vapor-phase boundary (dew point). Click and drag the black dot on either diagram and the bar chart shows the amounts of liquid (blue) and ...
Vapor-Liquid Equilibrium Data. The experimental data shown in these pages are freely available and have been published already in the DDB Explorer Edition.The data represent a small sub list of all available data in the Dortmund Data Bank.For more data or any further information please search the DDB or contact DDBST.. Explorer Edition Data Main Page
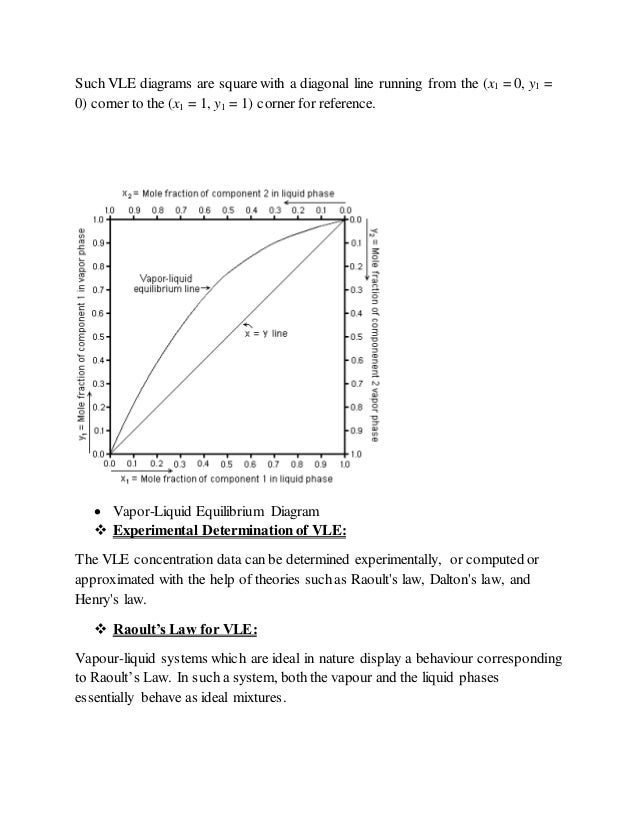
Vapor–liquid equilibrium diagrams — For each component in a binary mixture, one could make a vapor–liquid equilibrium diagram. Such a diagram would ...
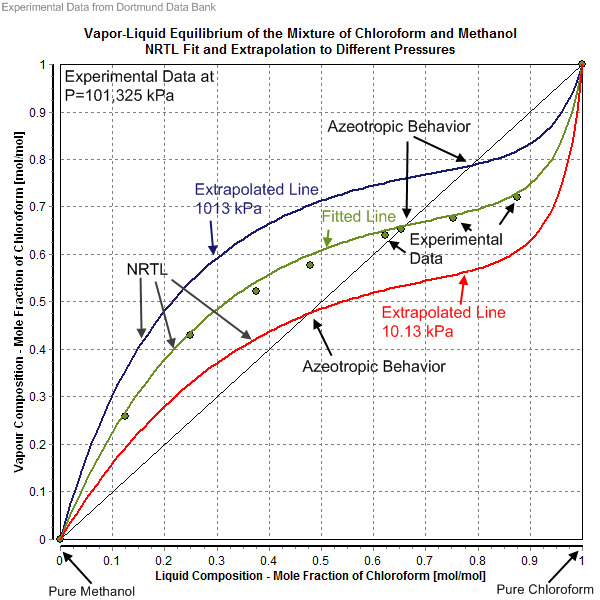
File Vapor Liquid Equilibrium Of The Mixture Of Chloroform And Methanol Nrtl Fit And Extrapolation To Different Pressures Png Wikimedia Commons
in the two-phase region. What saturated liquid and saturated vapour coexists in equilibrium? The P-x l and P-y 1 lines meet at the edges of the diagram what saturated liquid and saturated vapour of the pure species coexist ad the vapour pressures and .
Vapor-liquid equilibrium, abbreviated as VLE by some, is a condition where a liquid and its vapor (gas phase) are in equilibrium with each other, a condition or state where the rate of evaporation (liquid changing to vapor) equals the rate of condensation (vapor changing to liquid) on a molecular level such that there is no net (overall) vapor-liquid interconversion.
The equilibrium state corresponds to the saturated vapor pressure curve p = p s (T), which is limited by a triple point (T tr P tr) and a critical point (T c,p c) of the substance.(In the presence of surface tension forces at the phase interface, pressures values p (1) and p (g) in coexisting phases are not equal.) The Clausius-Clapeyron equation follows from Eq.
318 CHAPTER 10. Vapor/Liquid Eauilibrium: Introduction and KAC2, which represent the vapor pressure-vs.-T curves for pure species 1 and 2. More- over, the under and upper surfaces form a continuous rounded surface across the top of the diagram between Cl and C2, the critical points of pure species 1 and 2; the critical points of the

Vapor Liquid Equilibrium At 94 Kpa And Surface Tension At 298 15 K For Hexane Ethanol Cyclopentyl Methyl Ether Mixture Sciencedirect
liquid that is in equilibrium with vapor at differenttemperaturesdifferent temperatures. •In a binary system containing substances A ... Remembering that the x-y diagram is a plot of vapor (V) vs. liquid (L), it can be seen that the operating line is a simple y=mx+b equation. Therefore, with the
Liquid Vapor Equilibrium (LV) Objective The purpose of this experiment is to obtain the liquid-vapor phase diagram for methanol-ethyl acetate mixtures and to use this diagram to determine the boiling point and composition of the azeotropic mixture. Caution The liquids you are using are flammable. Watch out for burns. The still is very fragile. Do
Vapor-Liquid Equilibrium Diagram for Non-Ideal Mixtures. This simulation shows P-x-y and T-x-y diagrams for vapor-liquid equilibrium (VLE) of a benzene/ethanol mixture. This liquid mixture is non-ideal, and the system has an azeotrope (a condition where liquid and vapor have the same composition). The blue curve represents the liquid-phase ...
7 Vapor-Liquid Equilibria and Saturation Prof. Manolito E Bambase Jr. Department of Chemical Engineering. University of the Philippines Los Baños SLIDE 25 Vapor-Liquid Equilibrium for Multi-component Systems Consider a binary mixture with components A and B. Liquid X A0 X B0 Vapor Y A (P A) Y B (P B) Liquid X A X B Heating
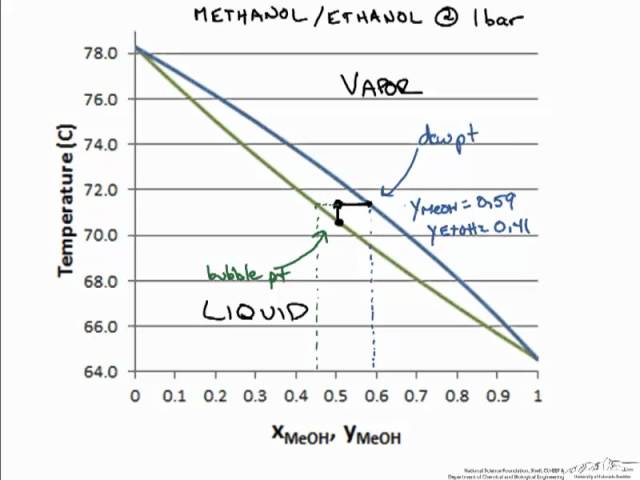
Two types of vapor-liquid equilibrium diagrams are widely used to represent data for two-component (binary) systems. The first is a "temperature versus x and y" diagram (Txy). The x term represents the liquid composition, usually expressed in terms of mole fraction. The y term represents the vapor composition. The second diagram is a plot of
on q or the liquid fraction of the feed. In the distillation literature, q is usually referred to as the thermal state of the feed. Note that q can be less than zero (superheated vapor) or greater than unity (subcooled liquid). The slope of the q-line is q/(q – 1). The construction of the two operating lines and the
The main objective of this practical is to determine the Vapour-Liquid Equilibrium for the given real system of two miscible liquids. it is also to construct the isobaric diagram and the equilibrium diagram of the two components. The other objective is to calculate the Raoul t's activity coefficients.
The vapor-liquid equilibrium relations for benzene (1)-toluene (2) at a total pressure of 200 kPa are given as a boiling-point Txy diagram shown in Figure 4.1-1. The upper curve is the saturated vapor curve (the dew-point curve) and the lower curve is the saturated liquid curve (the bubble-point curve).
Vapor-liquid equilibrium data for Ethanol-Water* n-Buianol-Wator# Ethyl Aeeiate-Water# and n-Hexane-Ethanol at atmospheric pr 00 sura and £or n-Hoxana-Sthanol at 2f50 mnu, 59$ mm.* 1270 mm*# Ijbj mm** 2J10 mm.#
Single Equilibrium Stages ()(1) [Ch.4][Ch. 4] •Phase Separation •Degree of Freedom Analysis-Gibbs phase rule F C P 2-General analysis •Binary Vapor-Liquid Systems-Examples of binary system-Phase equilibrium diagram-q-line-Phase diagram for constant relative volatility •AzeotropicSystems
Introduction To Chemical Engineering Processes Vapor Liquid Equilibrium Wikibooks Open Books For An Open World
The vapor-liquid equilibrium (VLE) behavior of a benzene (C 6 H 6)/toluene (C 7 H 8) mixture is demonstrated in P-x-y and T-x-y diagrams. The blue line represents the liquid-phase boundary (bubble point) and the green line represents the vapor-phase boundary line (dew point). Drag the black dot on either diagram and the bar chart shows the ...
In thermodynamics and chemical engineering, the vapor–liquid equilibrium (VLE) describes the distribution of a chemical species between the vapor phase and a liquid phase.. The concentration of a vapor in contact with its liquid, especially at equilibrium, is often expressed in terms of vapor pressure, which will be a partial pressure (a part of the total gas pressure) if any other gas(es ...
Txy Diagram. Txy diagram plots bubble and dew point curves at constant pressure P. Put down the liquid mol fraction x 1 from 0.0 to 1.0 with increment of 0.01 in spreadsheet. Iteration is done for each liquid mol fraction to estimate equilibrium temperature T and activity coefficient γ i.
Uses an interactive simulation to explain how to read a T-x-y diagram for a binary mixture that obeys Raoult's law. This simulation is located at: https://w...












0 Response to "34 vapour liquid equilibrium diagram"
Post a Comment