36 hydrogen energy level diagram colors
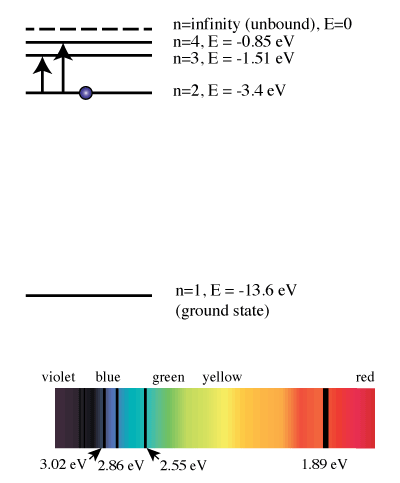
Chemistry. Chemistry questions and answers. Warm-up Question Match the six colors with the appropriate part of the hydrogen energy-level diagram to indicate the color of each emission. 1st attempt di See Periodic Table A Green B Ultraviolet Viole D) Indigo F Infrared. Energy level diagrams and the hydrogen atom. It's often helpful to draw a diagram showing the energy levels for the particular element you're interested in. The diagram for hydrogen is shown above. The n = 1 state is known as the ground state, while higher n states are known as excited states. If the electron in the atom makes a transition from ...
Layers of the Atmosphere Diagram 1. Give each student a copy of the following two pages. 2. Lay out 9” wide construction paper of various colors, plus rulers, scissors, and pencils. 3. Instruct students to measure and cut the following five 9” strips of paper, each in a …
Hydrogen energy level diagram colors
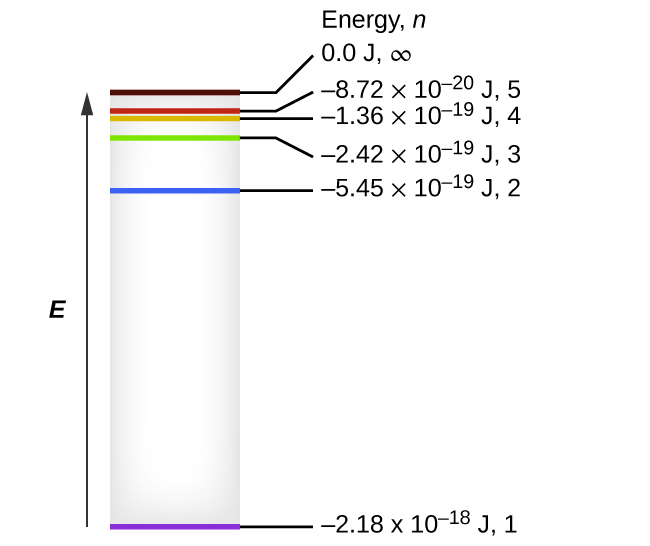
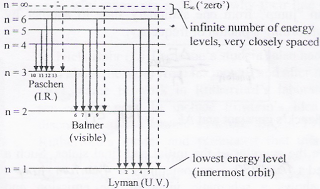
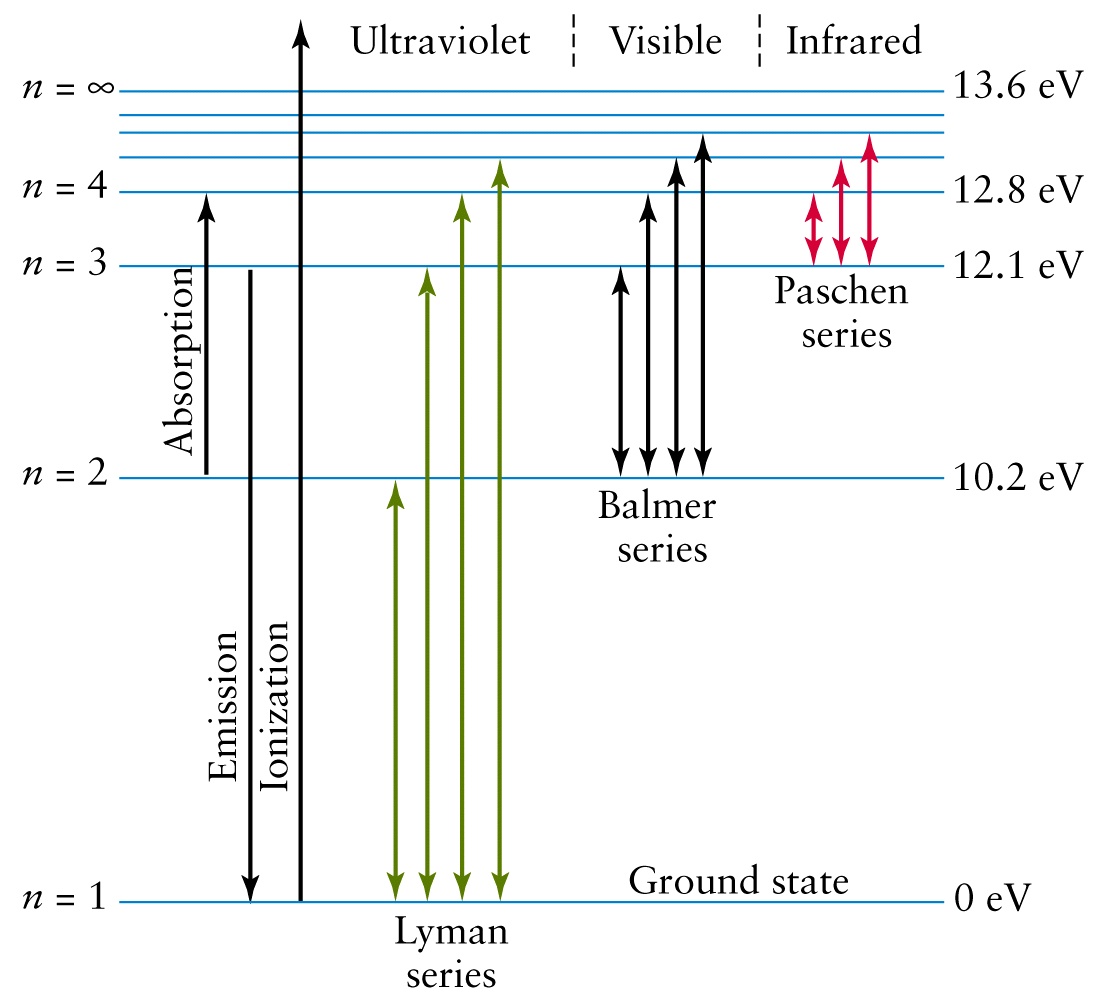
quantized. The diagrams below show a representation of how we can explain the line spectrum of hydrogen. When a photon is absorbed by a hydrogen atom, the energy of the photon causes the electron to undergo a transition to a higher energy level (n = 1 n = 2, for example). When a hydrogen atom PHYS 1493/1494/2699: Exp. 7 – Spectrum of the Hydrogen Atom 2 Introduction The physics behind: The spectrum of light The empirical Balmer series for Hydrogen The Bohr model (a taste of Quantum Mechanics) Brief review of diffraction The experiment: How to use the spectrometer and read the Vernier scale Part 1: Analysis of the Helium (He) spectrum The ionization energy of an atom is the energy required to remove the electron completely from the atom. (transition from ground state n = 0 to infinity n = ∞ ). For hydrogen, the ionization energy = 13.6eV. When an excited electron returns to a lower level, it loses an exact amount of energy by emitting a photon.
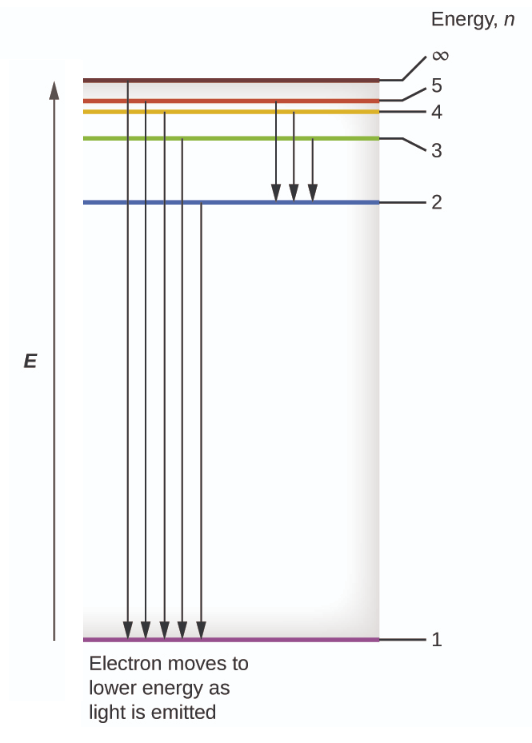
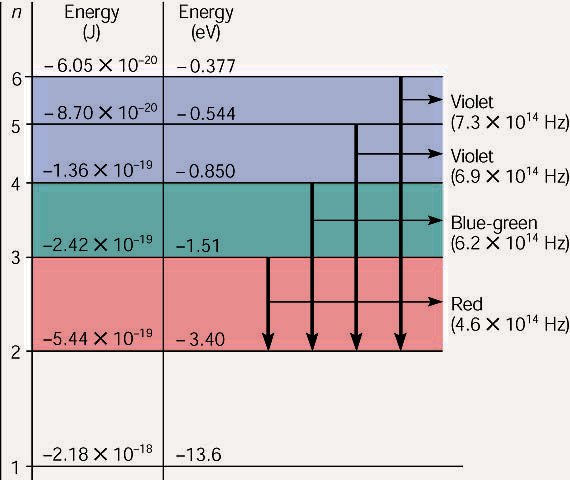
Hydrogen energy level diagram colors. Mar 31, 2005 · The electron in some hydrogen atoms may be excited into the n = 2 level. Other hydrogen atoms can have the electron excited into the n = 4 shell. Different elements emit different emission spectra when they are excited because each type of element has a unique energy shell or energy level system. The energy level diagram of the hydrogen atom is given below: The transition of electrons from a higher energy level (n>1) to the ground level (n=1) gives rise to the Lyman series in hydrogen spectra. where is the energy of the photon, is its frequency, and is Planck's constant.This concludes that only photons with specific energies are emitted by the atom. The principle of the atomic emission spectrum explains the varied colors in neon signs, as well as chemical flame test results (described below).. The frequencies of light that an atom can emit are dependent on states the electrons can ... If left undisturbed, our hydrogen atom likes to bind its electron as tightly as it can, and so we would find the electron in the lowest energy level, which is called the "ground state." However, if our atom is immersed in a beam of light from, say, a nearby star, sooner or later the atom will encounter a photon with an energy that is just the ...
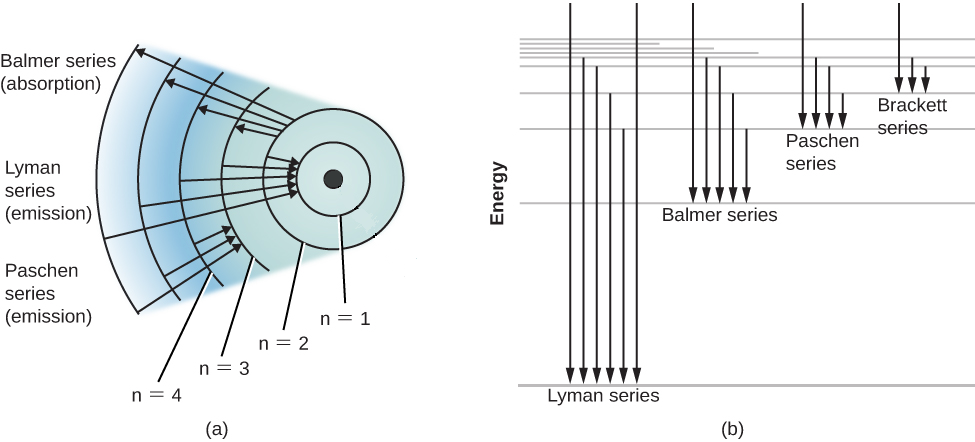
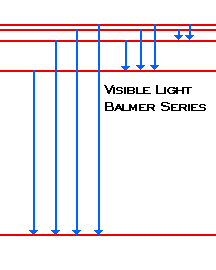
19. Identifr the drawing in Model 3 that depicts a hydrogen atom with an electron moving from energy level 5 to energy level 2. Refer to Models 1 and 2 for the following questions. a. Label the picture with "n=5 to n=2" and list the corresponding color of light emitted. … The Balmer series is characterized by the electron transitioning from n ≥ 3 to n = 2, where n refers to the radial quantum number or principal quantum number of the electron. The transitions are named sequentially by Greek letter: n = 3 to n = 2 is called H-α, 4 to 2 is H-β, 5 to 2 is H-γ, and 6 to 2 is H-δ.As the first spectral lines associated with this series are located in the ... Hydrogen has signature colors that appear when the atoms are excited. This image from NASA-Ames shows a hydrogen arc lamp fluorescing: Neils Bohr used the emission spectra of hydrogen to develop his model of the atom. In short, quantized energy levels release specific bands of light with unique colors. A description of the physics is here. Here ... The energy levels agree with the earlier Bohr model, and agree with experiment within a small fraction of an electron volt. If you look at the hydrogen energy levels at extremely high resolution, you do find evidence of some other small effects on the energy. The 2p level is split into a pair of lines by the spin-orbit effect.
What is energy level diagram? In chemistry, an electron shell, or energy level, may be imagined as an orbit with electrons around the nucleus of an atom. The closest shell to the nucleus is called the “K shell” followed by the “L shell” then the “M shell” and so on away from the nucleus. The shells can be denoted by alphabets (K, L ... Diagram of a typical plant, showing the inputs and outputs of the photosynthetic process. ... White light is separated into the different colors (=wavelengths) of light by passing it through a prism. Wavelength is defined as the distance from peak to peak (or trough to trough). ... is captured and modified by the addition of Hydrogen to form ... Energy level diagrams for IONS Atoms with 1, 2, or 3 valence electrons lose them to form 1+, 2+ or 3+ ions respectively. naming metallic ions - the full name of the atom is followed by the word ion. Mg2+ is the magnesium ion Group 1 (1+) (lose 1e) Group 2 (2+) (lose 2e) Group 13 (3+) (lose 3e) Energy level diagrams for IONS So, if you passed a current through a tube containing hydrogen gas, the electrons in the hydrogen atoms are going to absorb energy and jump up to a higher energy level. When those electrons fall down to a lower energy level they emit light and so we talked about …
Which Drawing Best Illustrates The Phenomenon Responsible For The Specific Color Emitted By An Element When It Is Vaporized Quora
So, very hot stars will have weak Balmer series hydrogen lines because most of their hydrogen has been ionized. Recall also that it takes energy to raise an electron from a lower level to a higher level. So, if the cloud of gas is too cool, the electrons will all be in …
The ionization energy of an atom is the energy required to remove the electron completely from the atom. (transition from ground state n = 0 to infinity n = ∞ ). For hydrogen, the ionization energy = 13.6eV. When an excited electron returns to a lower level, it loses an exact amount of energy by emitting a photon.

The Energy Diagram Below Shows The Absorption Emission Spectrum For Hydrogen What Is The Smallest Energy Possible Of An Absorbed Photon A 0 66 Ev B 0 85 Ev C 3 4 Ev D 10 2 Ev
PHYS 1493/1494/2699: Exp. 7 – Spectrum of the Hydrogen Atom 2 Introduction The physics behind: The spectrum of light The empirical Balmer series for Hydrogen The Bohr model (a taste of Quantum Mechanics) Brief review of diffraction The experiment: How to use the spectrometer and read the Vernier scale Part 1: Analysis of the Helium (He) spectrum
quantized. The diagrams below show a representation of how we can explain the line spectrum of hydrogen. When a photon is absorbed by a hydrogen atom, the energy of the photon causes the electron to undergo a transition to a higher energy level (n = 1 n = 2, for example). When a hydrogen atom

Solved The Following Is An Energy Level Diagram For Electronic Transitions In The Bohr Hydrogen Atom A Explain Why The Energy Levels Get Closer Together As They Increase Provide Mathematical Support For This B
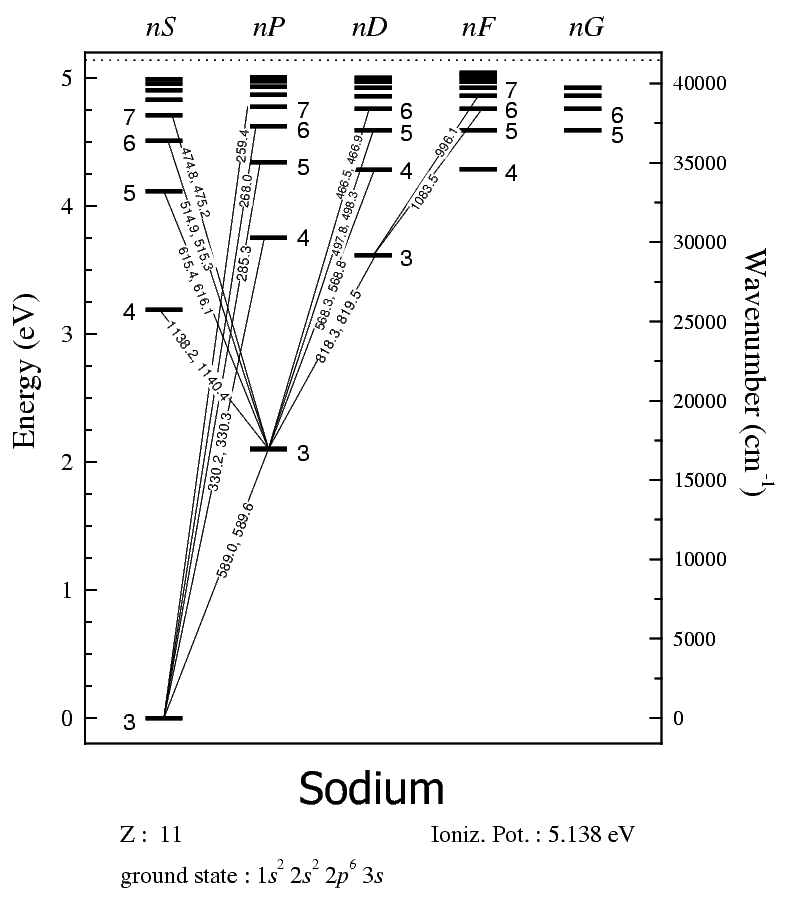
Why Do Certain Atoms Emit Certain Colors Of Light How Do They Get The Energy To Do So And What Kinds Of Particles Are Involved In This Process Socratic
















0 Response to "36 hydrogen energy level diagram colors"
Post a Comment