36 liquid vapor phase diagram
1.2 BINARY VLE PHASE DIAGRAMS Two types of vapor–liquid equilibrium diagrams are widely used to represent data for two-component (binary) systems. The first is a “temperature versus x and y” diagram (Txy). The x term represents the liquid composition, usually expressed in terms of mole fraction. The y term represents the vapor composition. 5.8 Liquid-Liquid phase diagrams Key points 1. Phase separation of partially miscible liquids may occur when T is below UCST or above LCST 2. UCST is the highest T at which phase separation occurs. LCST is the lowest T at which phase separation occurs 3. The outcome of a distillation of a low-boiling ...
by GA Chapela · 2013 · Cited by 6 — The liquid-vapor phase diagrams of equal size diameter σ binary mixtures of screened potentials have been reported for several ranges of interaction using ...
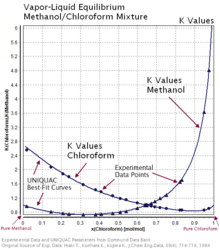
Liquid vapor phase diagram
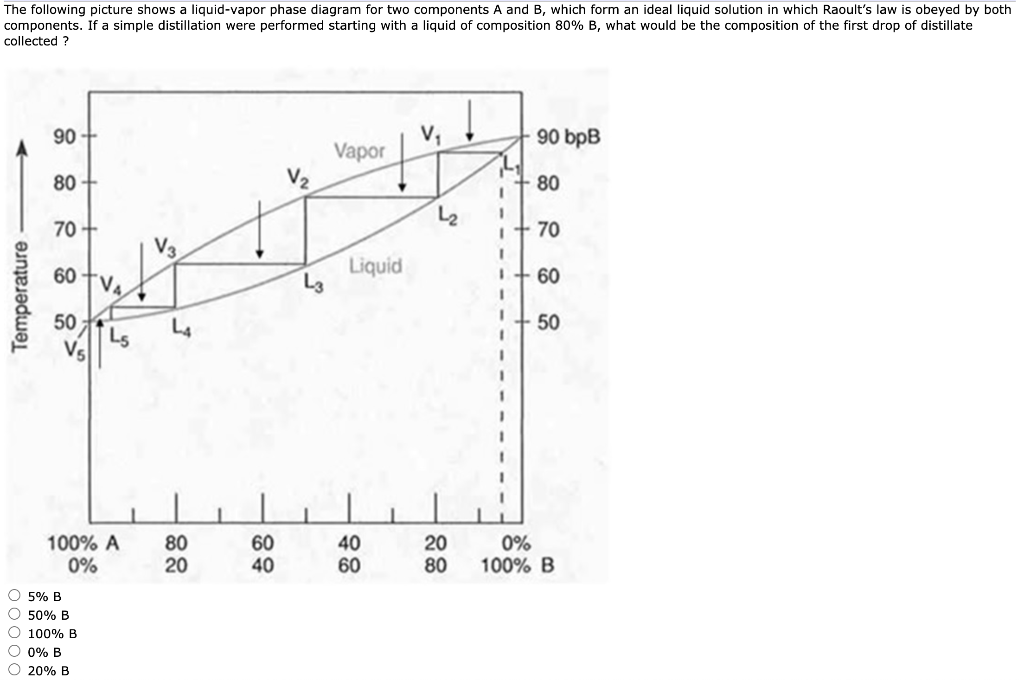
A liquid-vapor phase diagram of a binary system can be constructed by using a reflux apparatus. When a mixture of two soluble liquids is heated to a boiling point, the vapor phase is condensed and trapped in the pocket below the condenser. Under equilibrium conditions, the trapped condensate represents the vapor phase while the liquid remaining ... Construct a phase diagram using temperature versus mole percent composition. In the present case, there will be two compositions plotted at each equilibrium ...12 pages Phase Determination (Case 1 Cont’d) • Case 1: Given P and T – Look up saturation table – Compare given P and T against saturation values in the table ¾In temperature table, Recall constant temperature line on P-v diagram If P > P sat(T), compressed liquid. If P = P sat(T), saturated liquid-vapor mixture. If P < P sat(T), superheated vapor.
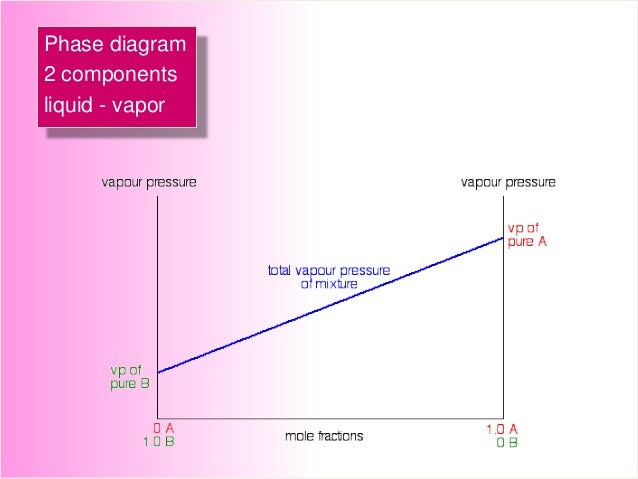
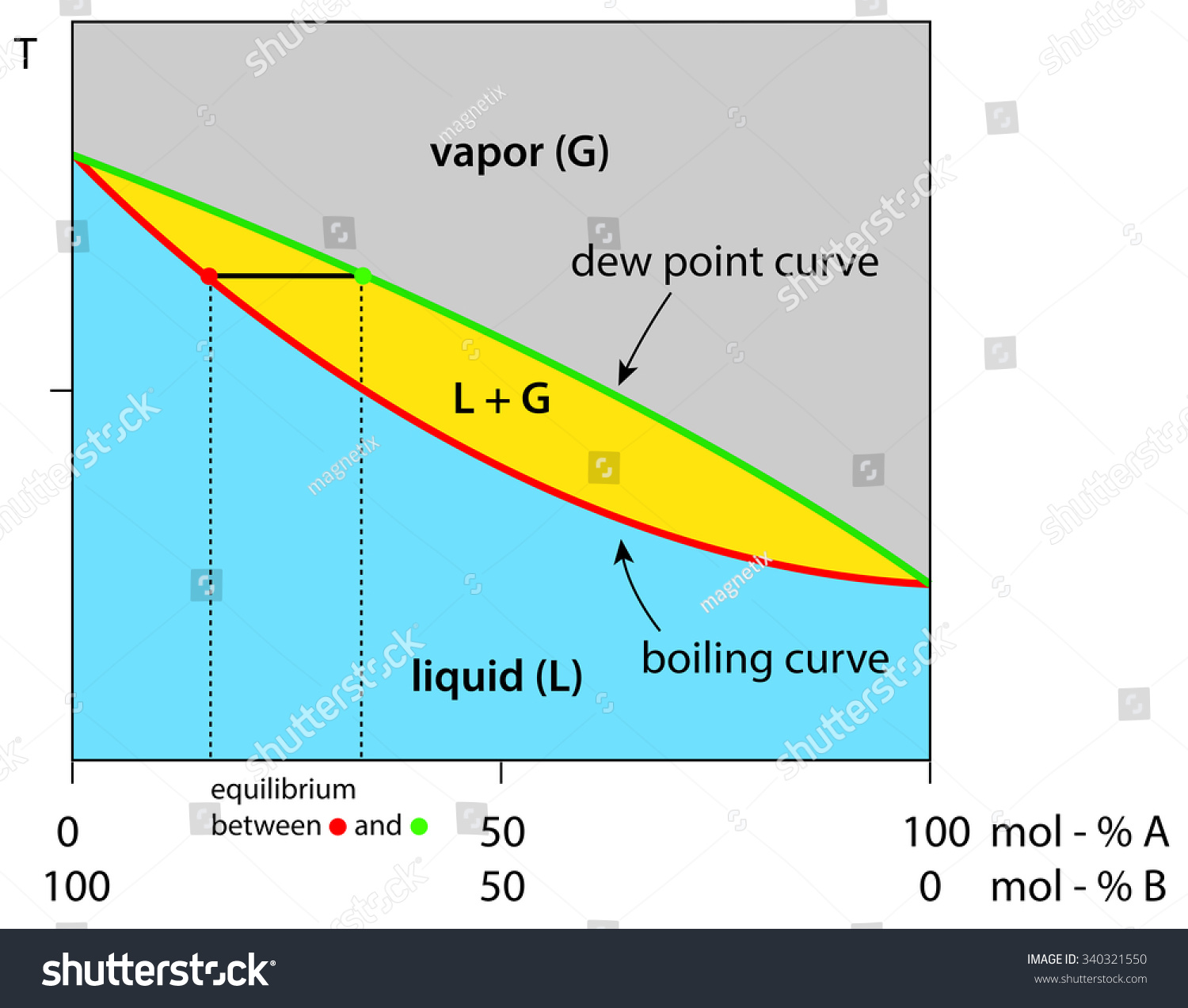
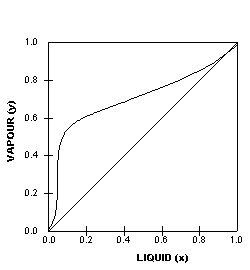
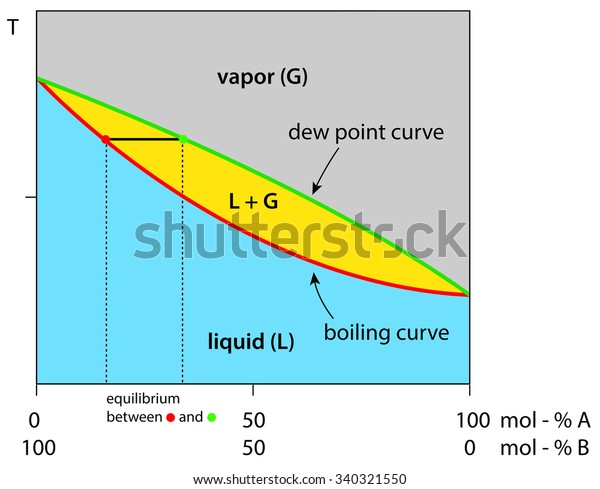
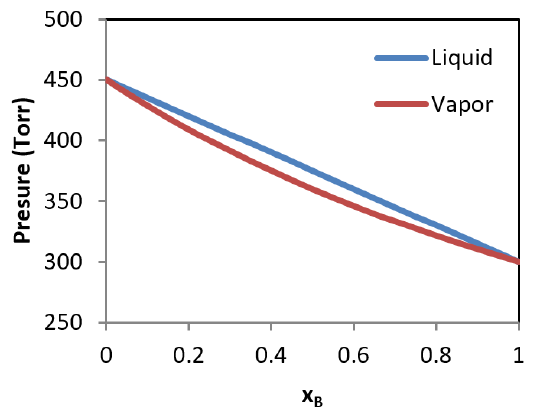
Liquid vapor phase diagram. A liquid-vapor phase diagram plots temperature of mixture versus the mole fraction or composition. A phase diagram of 2 components that will have differing molecular interactions causing mixture’s boiling point to rise/drop will show an azeotrope, in which there will be 2 lobes that either concave down/up depending on if there’s positive ... Chapter 8 Phase Diagrams. (b) The interpretation of diagrams. Point a represents the vapor pressure of a mixture with liquid composition xA and b represents the composition of the vapor that is in equilibrium with the liquid at that pressure. Note that when two phases are in equilibrium, P = 2, so F’ = 1. Vapor Liquid Phase Diagrams. Vapor-liquid equilibrium data may be obtained by experiment, by thermodynamic calculation, or in published sources. It is typically presented either in tabular form or as an equilbrium diagram. Diagrams may take several forms: boiling point diagrams Txy, Pxy diagrams ternary diagrams solubility diagrams In thermodynamics and chemical engineering, the vapor–liquid equilibrium (VLE) describes the distribution of a chemical species between the vapor phase and a liquid phase.. The concentration of a vapor in contact with its liquid, especially at equilibrium, is often expressed in terms of vapor pressure, which will be a partial pressure (a part of the total gas pressure) if any other gas(es ...
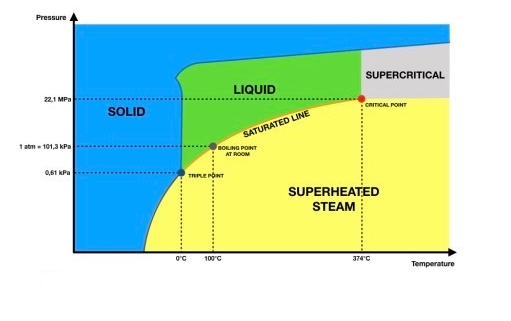
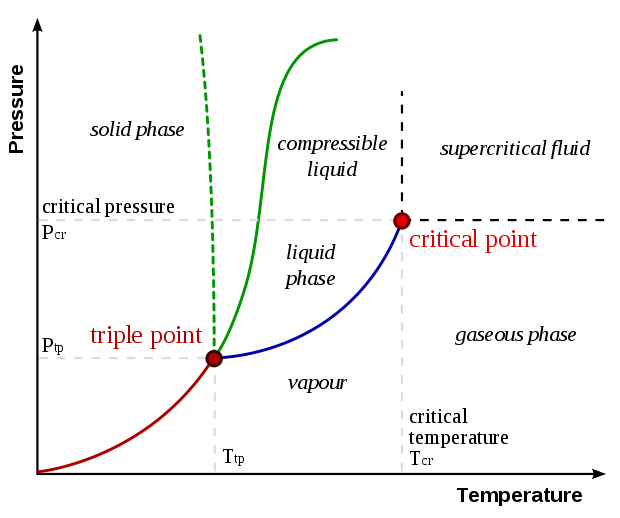
8 Mar 2021 — Oftentimes, it is desirable to depict the phase diagram at a single pressure so that temperature and composition are the variables included in ... the vapor phase is a saturated vapor. Liquid water continues to evaporate with additional heat until it becomes all saturated vapor at point (g). Any further heating will cause an increase in both temperature and specific volume and the saturated vapor becomes superheated vapor denoted by point (s) in Figure 3.2-2. For a two-phase liquid-vapor ... by P Schienbein · 2017 · Cited by 25 — On the basis of ab initio Gibbs ensemble Monte Carlo simulations, we map the liquid–vapor phase diagram of water described by the RPBE ... Two-phase dome Superheated vapor Comp. liquid S a t u rat ed v a p o r l i n e S a t u r a t e d l i q u i d l i n e Critical Point Vapor Liquid Solid S u b l i m a t i o n V a p o r i z a t i o M n e l t i n g Critical Point Critical State and Reduced Coordinate • Recall the phase diagrams of a general substance: cr R P P reduced pressure: P ...
Phase Determination (Case 1 Cont’d) • Case 1: Given P and T – Look up saturation table – Compare given P and T against saturation values in the table ¾In temperature table, Recall constant temperature line on P-v diagram If P > P sat(T), compressed liquid. If P = P sat(T), saturated liquid-vapor mixture. If P < P sat(T), superheated vapor. Construct a phase diagram using temperature versus mole percent composition. In the present case, there will be two compositions plotted at each equilibrium ...12 pages A liquid-vapor phase diagram of a binary system can be constructed by using a reflux apparatus. When a mixture of two soluble liquids is heated to a boiling point, the vapor phase is condensed and trapped in the pocket below the condenser. Under equilibrium conditions, the trapped condensate represents the vapor phase while the liquid remaining ...

Phase Equilibrium And Intermolecular Forces Concept Development Studies In Chemistry 2013 Openstax Cnx
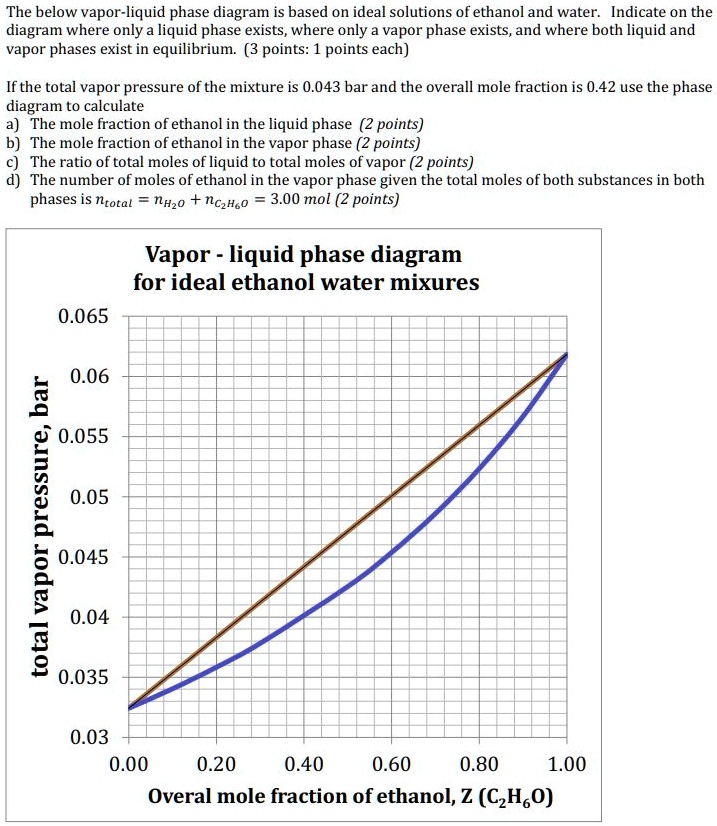
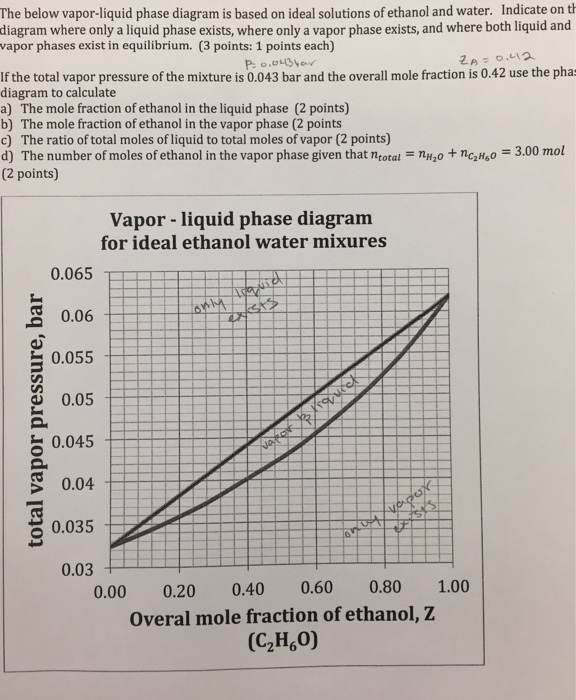
Solved The Below Vapor Liquid Phase Diagram Is Based On Ideal Solutions Of Ethanol And Water Indicate On The Diagram Where Only A Liquid Phase Exists Where Only Vapor Phase Exists And Where Both

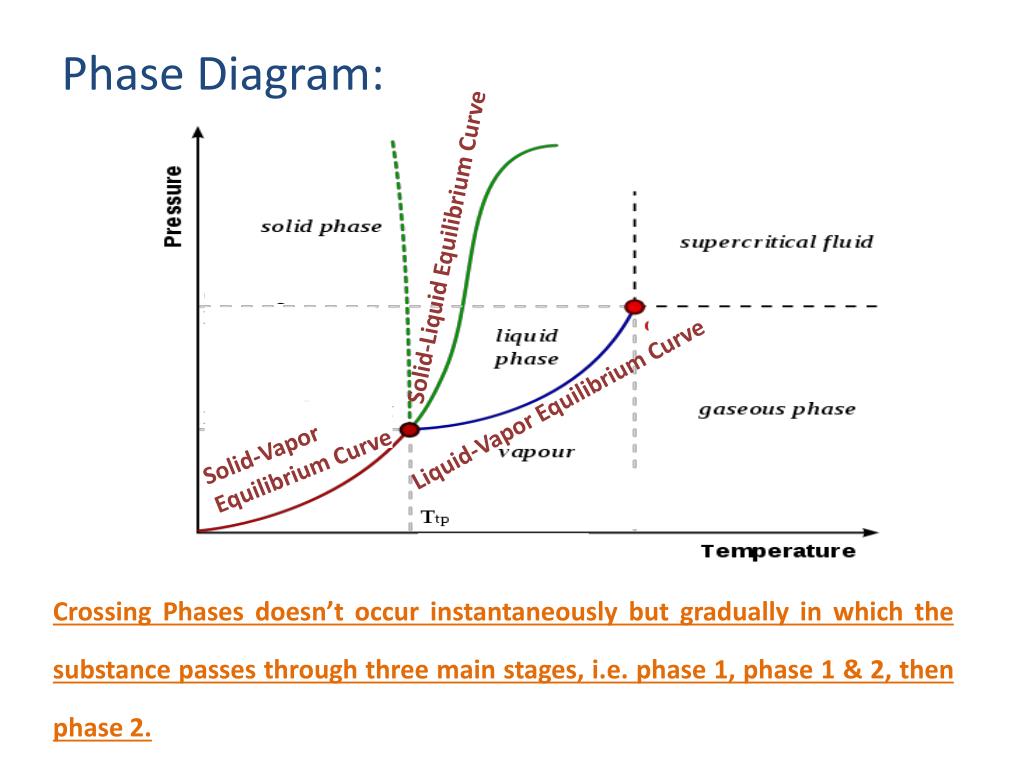



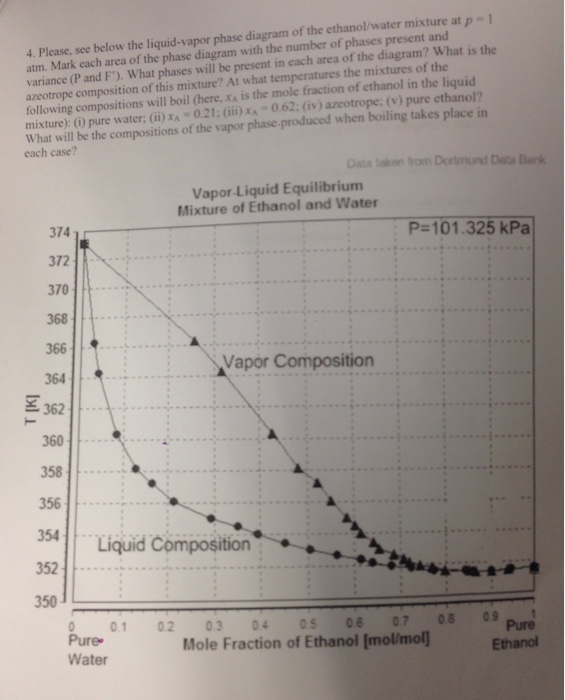











0 Response to "36 liquid vapor phase diagram"
Post a Comment