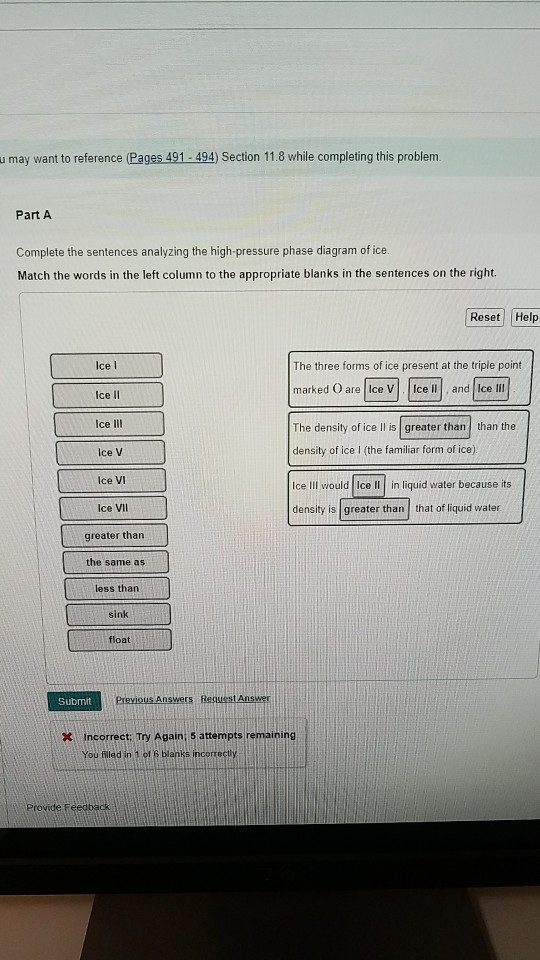
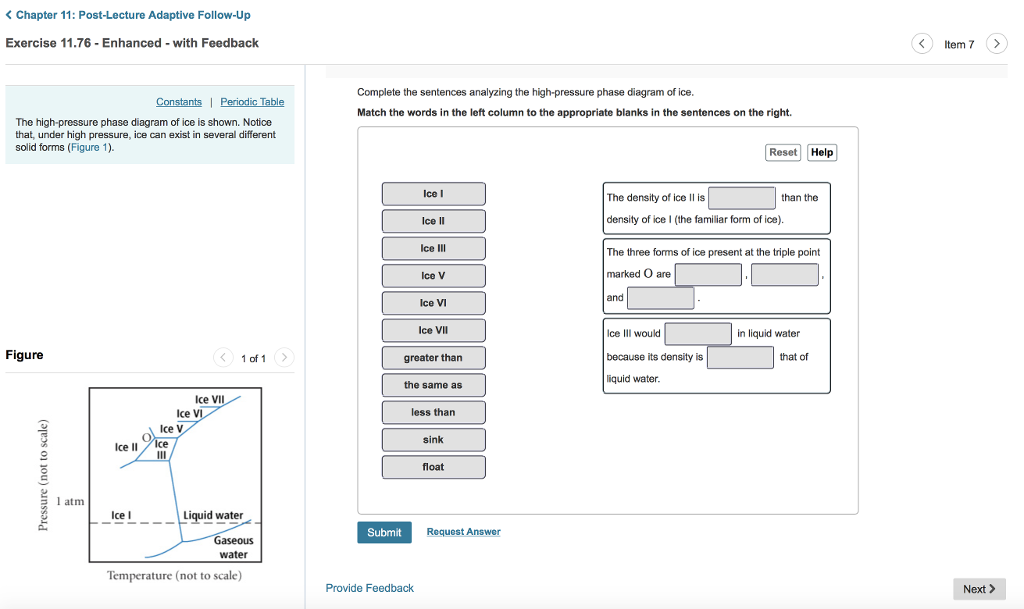
39 complete the sentences analyzing the high-pressure phase diagram of ice.
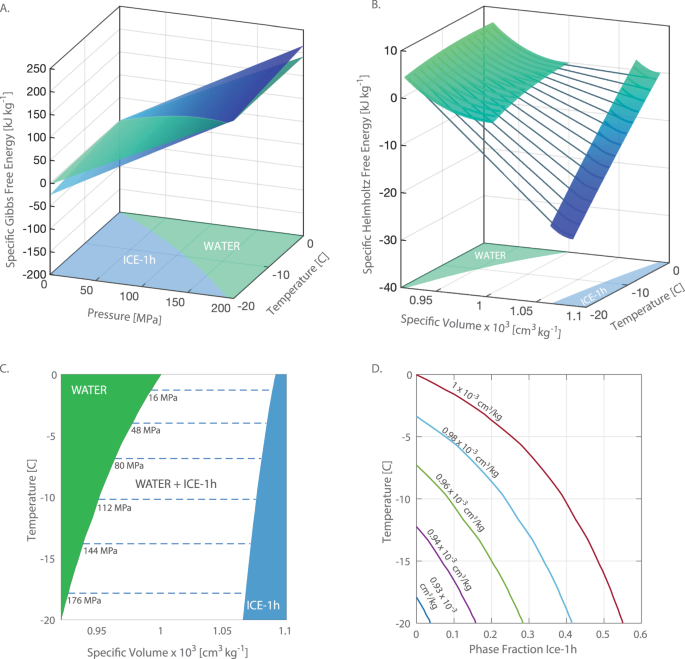
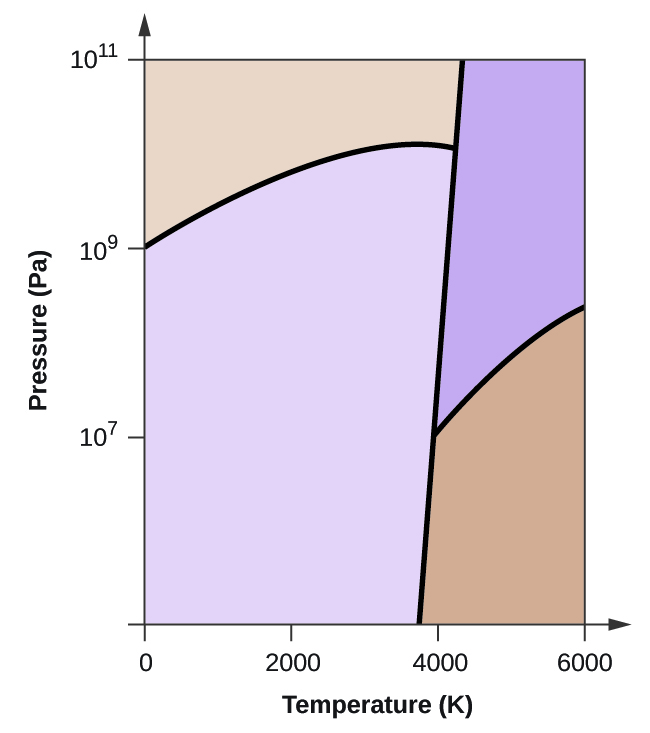
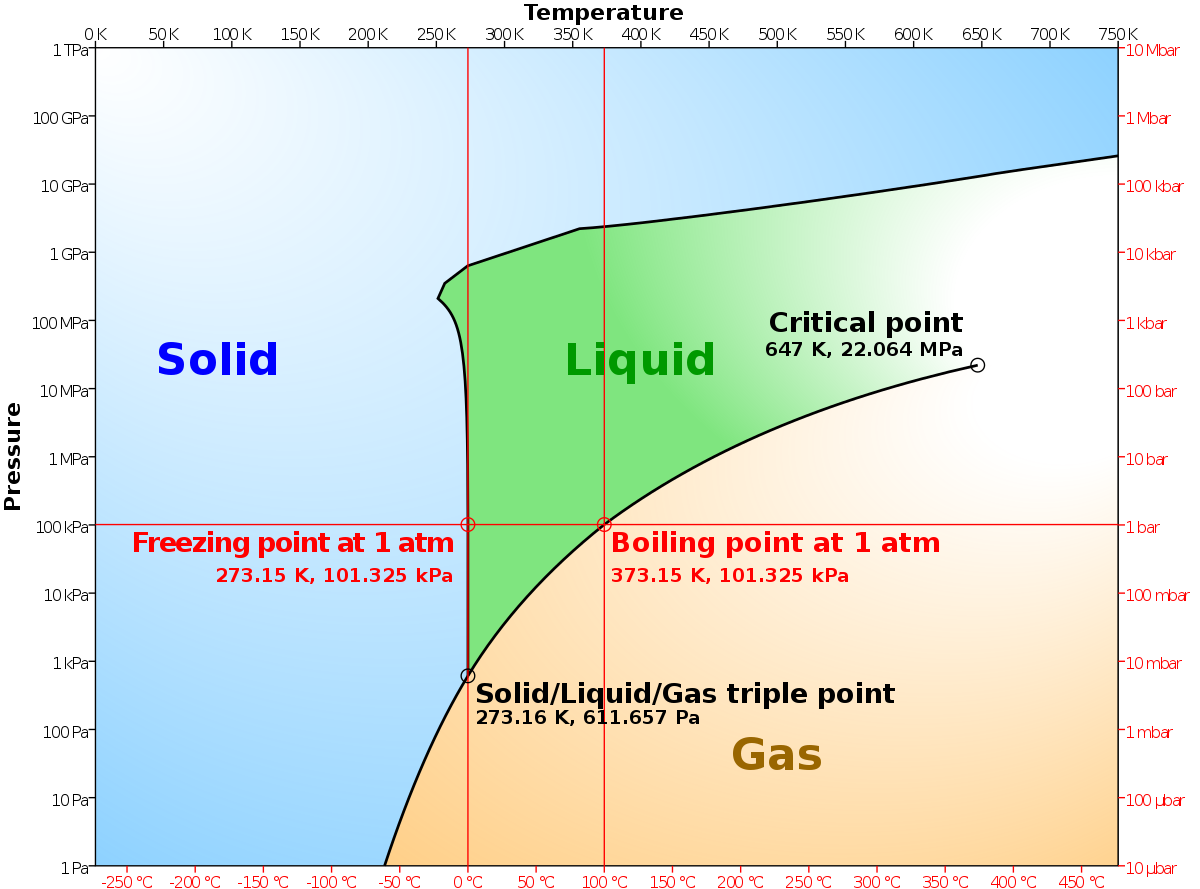
Several aspects of the phase diagram of water at high pressure are immensely controversial: the location of the melting line 5,10,11,12,13,14,15,16,17,18 and the existence, structure, physical ... The critical point and the orange line in the ice-one phase space refer to the low-density (LDA) and high-density (HDA) forms of amorphous water (ice) . Although generally accepted, the existence of this second, if metastable, critical point is impossible to prove at the present time and is disputed by some [ 200 , 618 , 628 ].
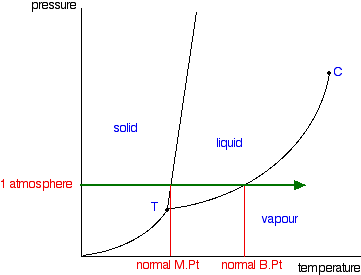
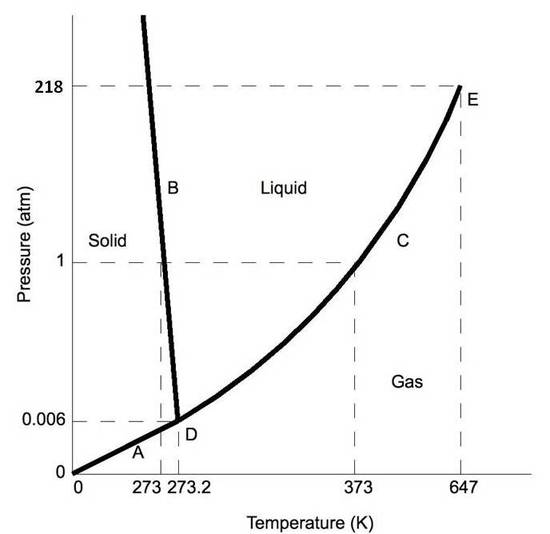
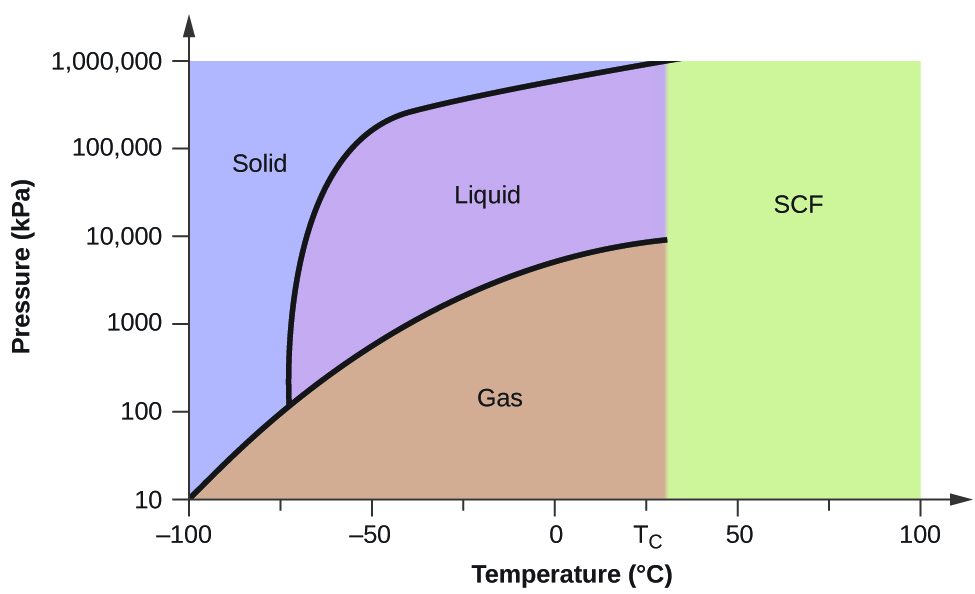
We can use the phase diagram to identify the physical state of a sample of water under specified conditions of pressure and temperature. For example, a pressure of 50 kPa and a temperature of −10 °C correspond to the region of the diagram labeled “ice.”. Under these conditions, water exists only as a solid (ice).
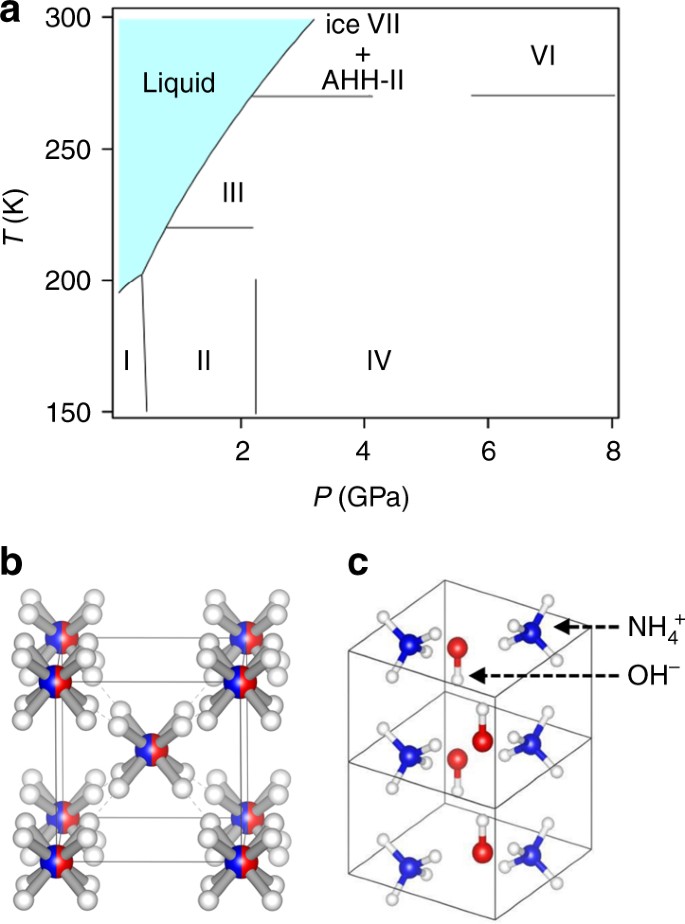
Complete the sentences analyzing the high-pressure phase diagram of ice.
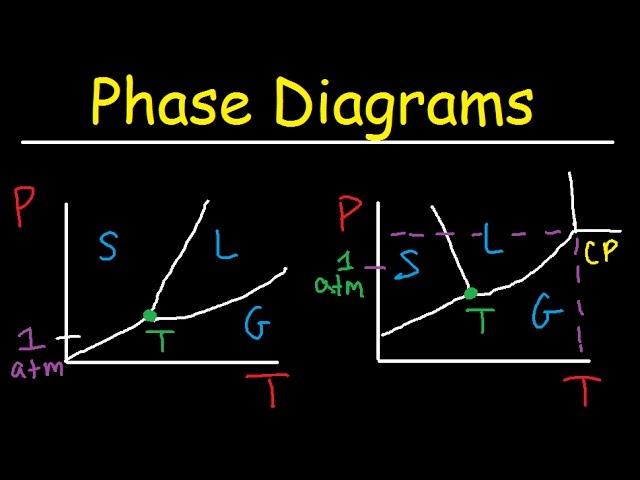
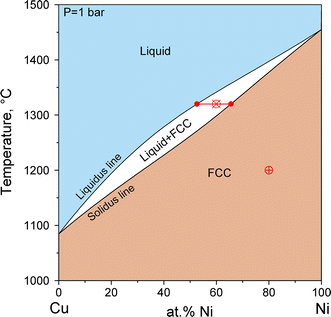
Therefore, a pressure change has the opposite effect on those two phases. If ice is relatively near its melting point, it can be changed into liquid water by the application of pressure. The water molecules are actually closer together in the liquid phase than they are in the solid phase. Refer again to water’s phase diagram ( Figure above ). Phase Diagrams • Indicate phases as function of T, Co, and P. • For this course:-binary systems: just 2 components.-independent variables: T and Co (P = 1 atm is almost always used). • Phase Diagram for Cu-Ni system Adapted from Fig. 9.3(a), Callister 7e. (Fig. 9.3(a) is adapted from Phase Diagrams of Binary Nickel Alloys , P. Nash J. Dubessy, M.-C. Caumon , F. Rull · 2012 · Sciencetransformations induced by pressure and temperature in the water structure and in the hydrogen bonding interaction is the water/ice phase diagram, ...
Complete the sentences analyzing the high-pressure phase diagram of ice.. this's Jumper eleven Section Problems seventy six. The high pressure phase diagram of Isis shown here I've done the best I could to try to represent this out of a textbook note that under high pressure ice can exist in several different ice solid forms when three wish Three forms of ice are present at the triple point marked Oh, and it's The book points to where I've indicated rate. Question: u may want to reference (Pages 491-494) Section 11.8 while completing this problem Part A Complete the sentences analyzing the high-pressure phase diagram of ice. Match the words in the left column to the appropriate blanks in the sentences on the right. Reset Help Ice l Ice II ice!" Ice V Ice VI Ice VII The three forms of ice present ... A phase diagram is a temperature-pressure plot that summarizes the conditions under which a substance exists as a solid, liquid, or gas. The curves that separate the phases are known as phase boundaries. Each phase boundary represents the equilibrium between the phases on either side of the curve. Identify the components of the phase diagram of ... 28 Aug 2015 — At high pressures, the properties of superionic ice are largely unknown. ... Here we report a comprehensive phase diagram of SI ice.
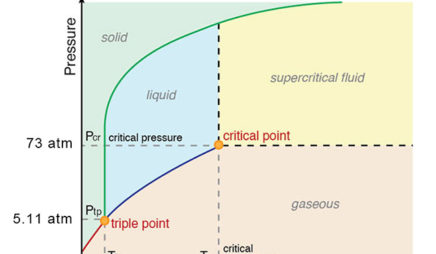
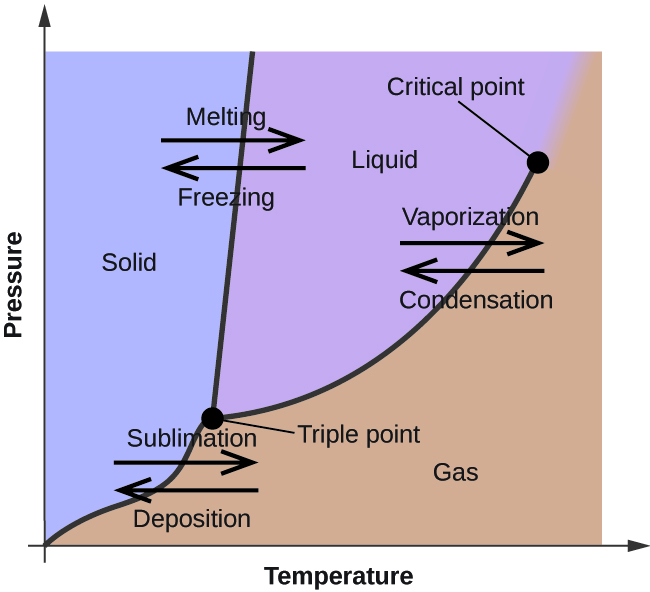
Patrick Echlin · 1992 · ScienceA phase diagram of many of these different forms in relation to the liquid state is ... The high pressures push the water molecules closer together and the ... Phase Diagrams. The figure below shows an example of a phase diagram, which summarizes the effect of temperature and pressure on a substance in a closed container. Every point in this diagram represents a possible combination of temperature and pressure for the system. The diagram is divided into three areas, which represent the solid, liquid ... Transcribed image text: Constants 1 Periodic Table hat, under high pressure, ice can exist in several different solid forms (Figure 1). lce I n liquid water because its density is Figure 1 of 1 Ice III familiar form of ice). Ice lce VI lce VIl greater than Ice Ice The three forms of ice present at the triple point marked O are and Ice lI 1 atm water float Temperature (not to scale) Notice that, under high pressure, ice can exist in several different solid forms.Would ice III sink or float in liquid water? FREE Expert Solution. We have to ...1 answer · Top answer: We have to determine if ice III would sink or float on liquid water.Recall that substances of lower density float over substances of highest density.A substance ...
J. Dubessy, M.-C. Caumon , F. Rull · 2012 · Sciencetransformations induced by pressure and temperature in the water structure and in the hydrogen bonding interaction is the water/ice phase diagram, ... Phase Diagrams • Indicate phases as function of T, Co, and P. • For this course:-binary systems: just 2 components.-independent variables: T and Co (P = 1 atm is almost always used). • Phase Diagram for Cu-Ni system Adapted from Fig. 9.3(a), Callister 7e. (Fig. 9.3(a) is adapted from Phase Diagrams of Binary Nickel Alloys , P. Nash Therefore, a pressure change has the opposite effect on those two phases. If ice is relatively near its melting point, it can be changed into liquid water by the application of pressure. The water molecules are actually closer together in the liquid phase than they are in the solid phase. Refer again to water’s phase diagram ( Figure above ).

Novel Color Center Platforms Enabling Fundamental Scientific Discovery Norman 2021 Infomat Wiley Online Library
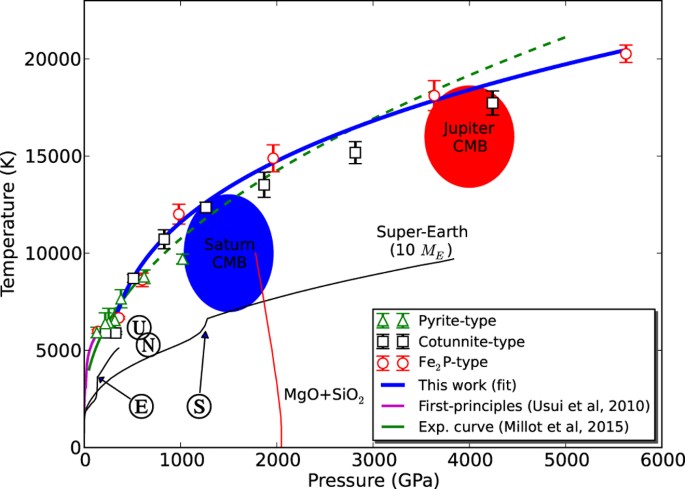
Melting Curve Of Sio2 At Multimegabar Pressures Implications For Gas Giants And Super Earths Scientific Reports
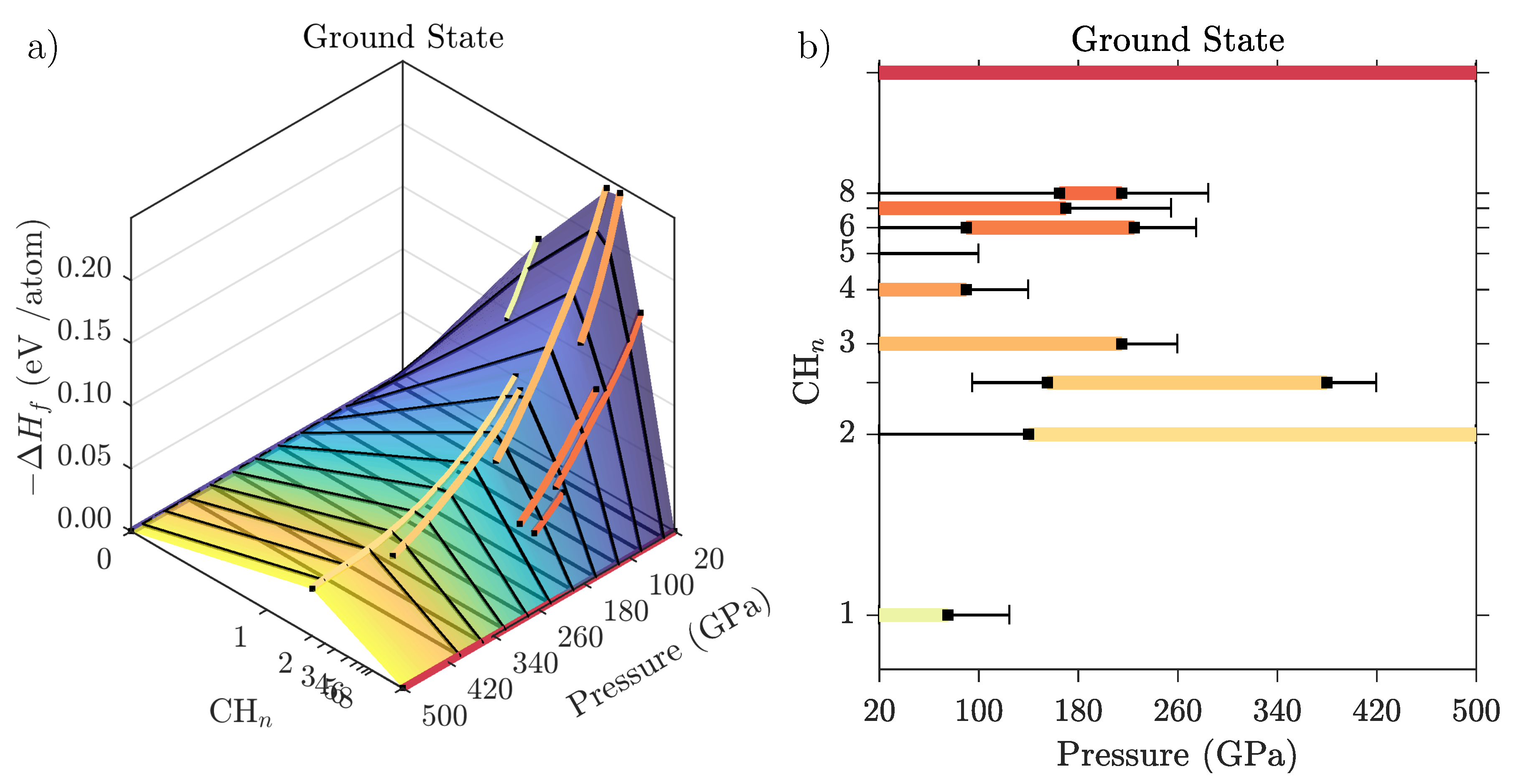
Geosciences Free Full Text High Pressure Hydrocarbons Revisited From Van Der Waals Compounds To Diamond Html
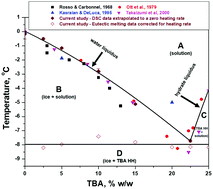
A Refined Phase Diagram Of The Tert Butanol Water System And Implications On Lyophilization Process Optimization Of Pharmaceuticals Physical Chemistry Chemical Physics Rsc Publishing
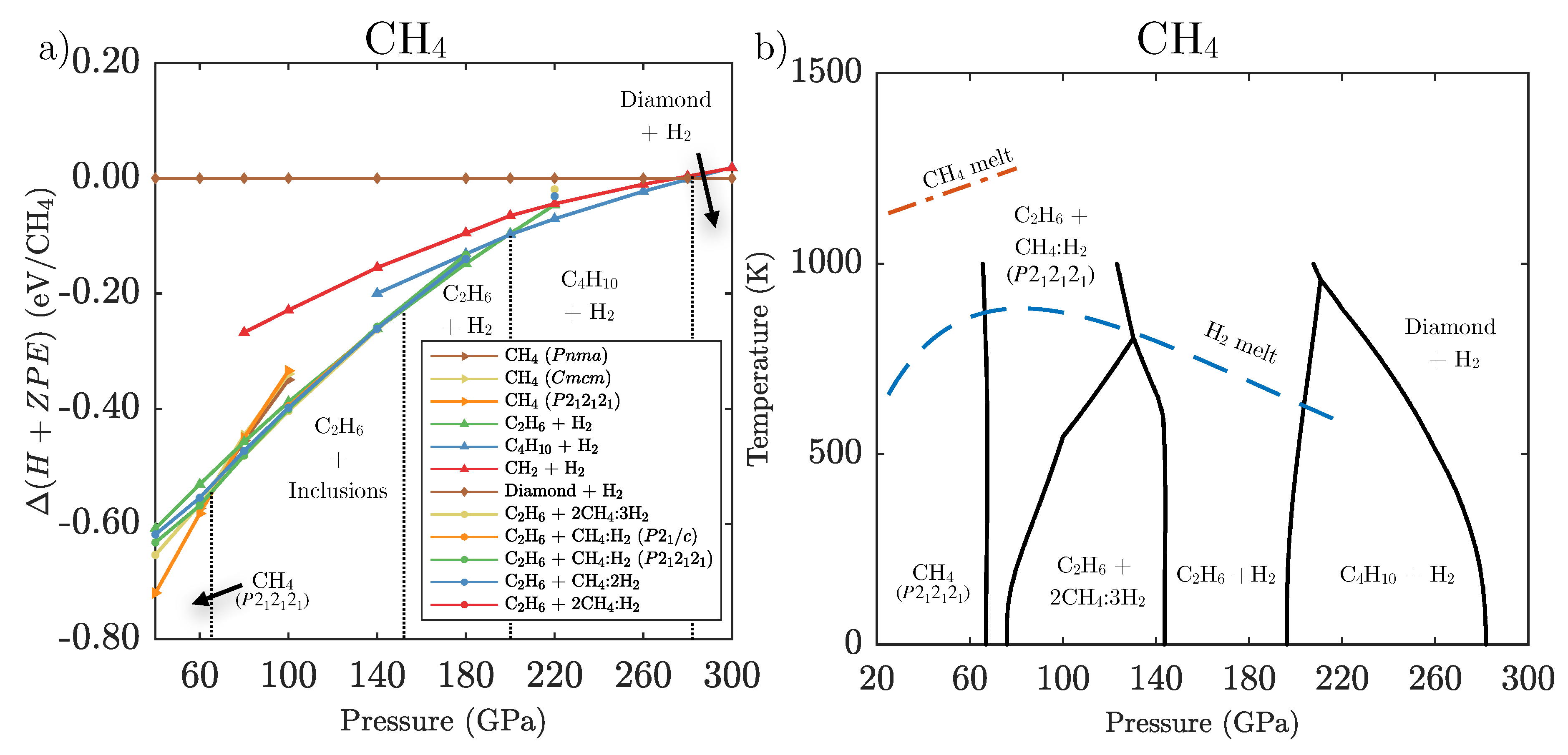
Geosciences Free Full Text High Pressure Hydrocarbons Revisited From Van Der Waals Compounds To Diamond Html
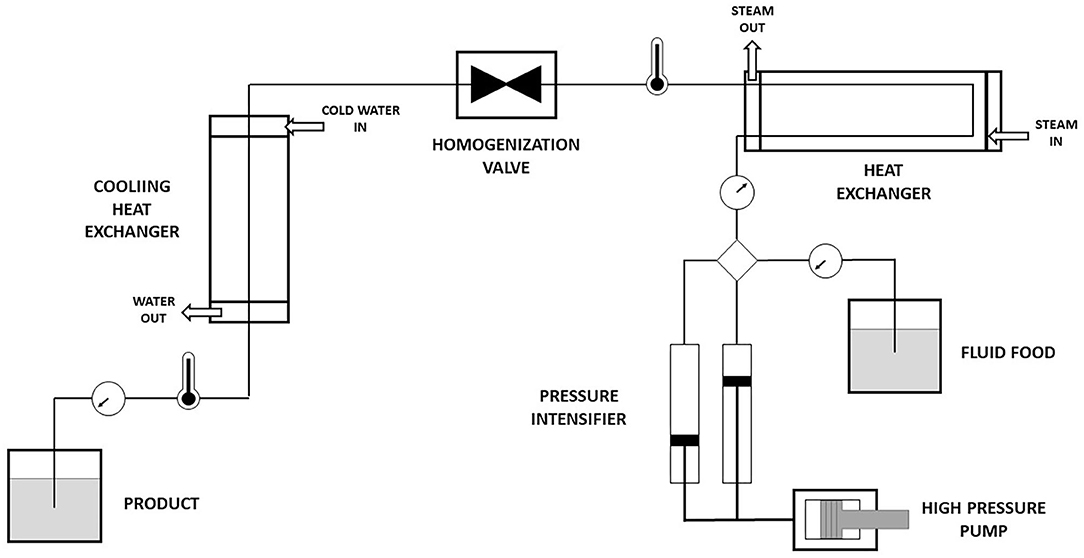
Frontiers Impact Of High Pressure Homogenization On The Extractability And Stability Of Phytochemicals Sustainable Food Systems

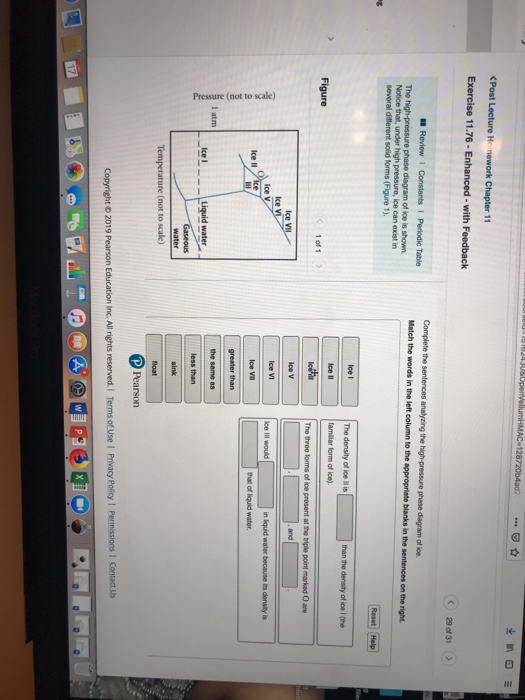










0 Response to "39 complete the sentences analyzing the high-pressure phase diagram of ice."
Post a Comment