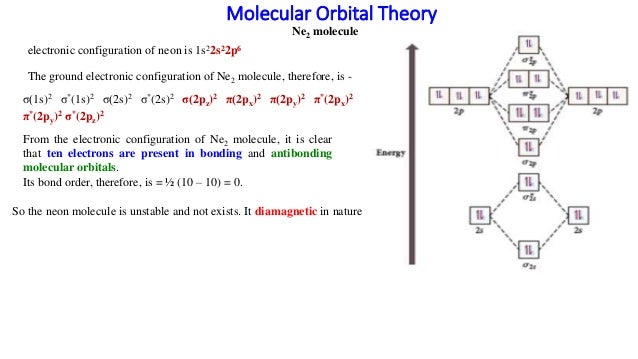
34 ne2 molecular orbital diagram
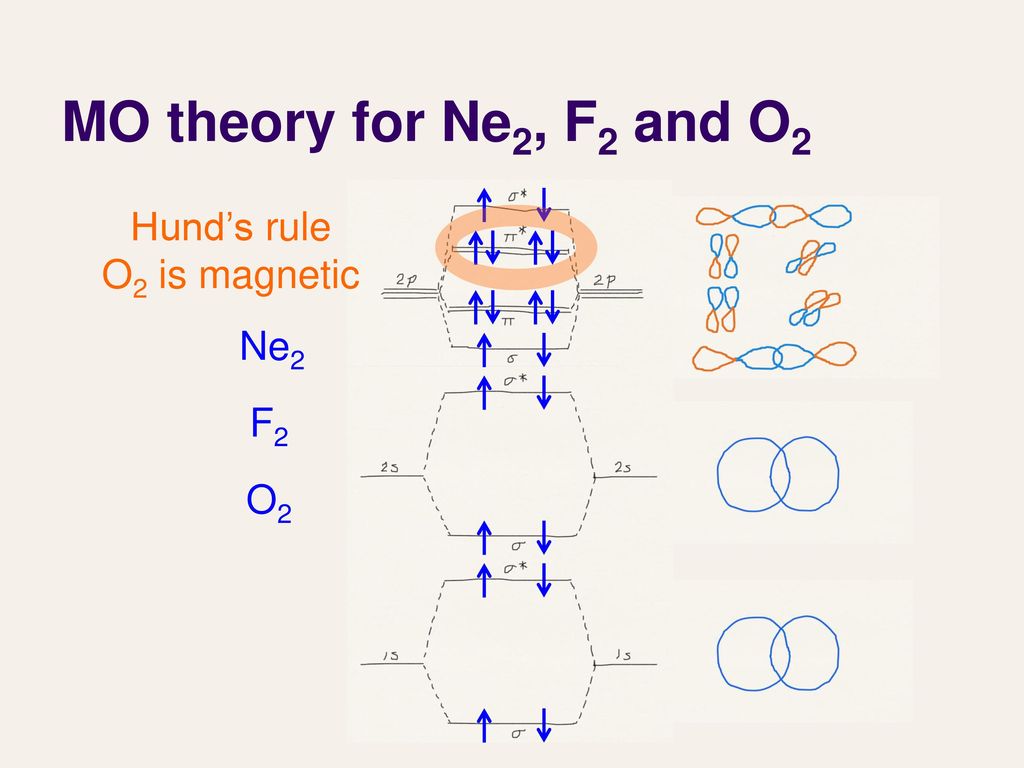
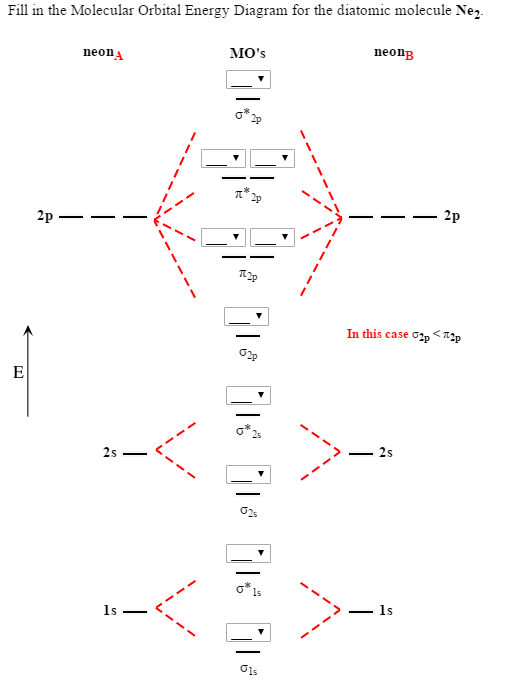
Problem: Determine the bond order from the molecular orbital diagram of N2, F2, and Ne2.Does the bond order calculated agree with what you would draw for the Lewis structures of these molecules? Explain your answer here in addition to providing the bond order values. For Ne2, construct three molecular orbital diagrams, one each for the neutral molecule, the +1 cation, and the -1 anion. Give each MO an appropriate label. Determine the electron configuration and bond order for each, and rank the three species in order of increasing bond order. Rationalize the trend in bond order in terms of bond strength.
0:21 Molecular Orbital Diagram of Oxygen Molecule3:30 Molecular Orbital Diagram of Florine Molecule5:25 Molecular Orbital Diagram of Neon MoleculeSo as we d...
Ne2 molecular orbital diagram
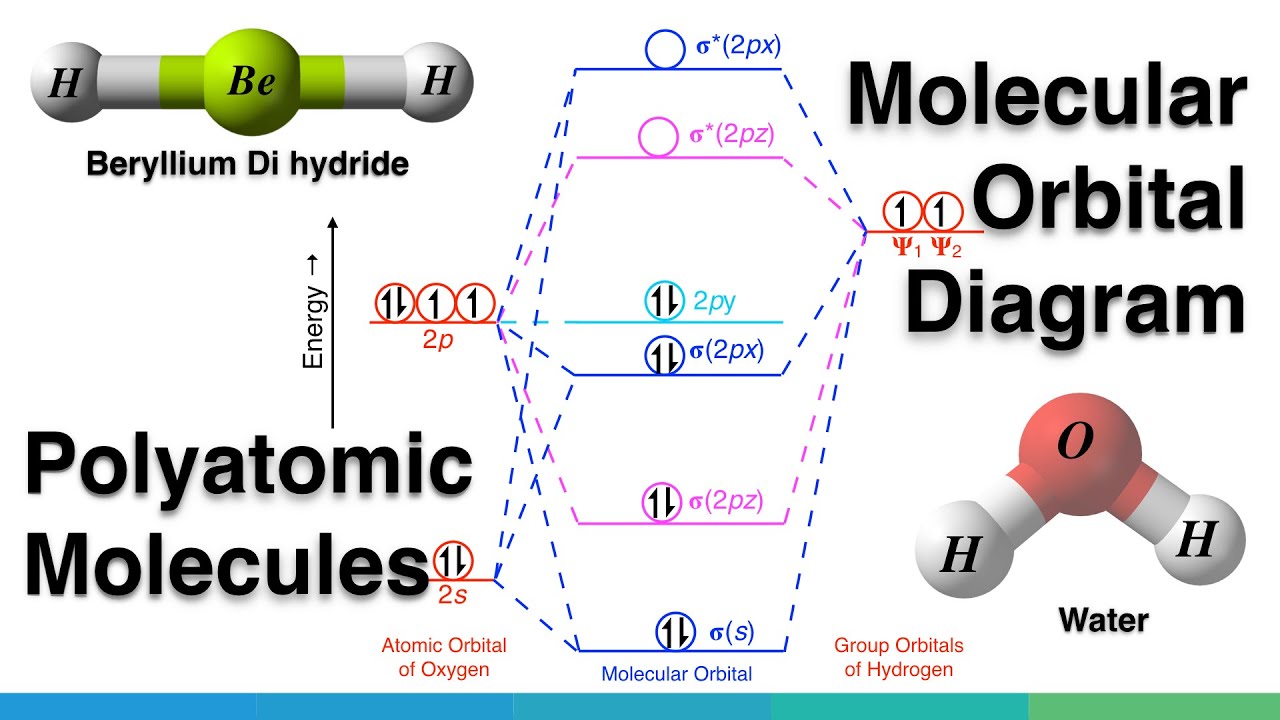
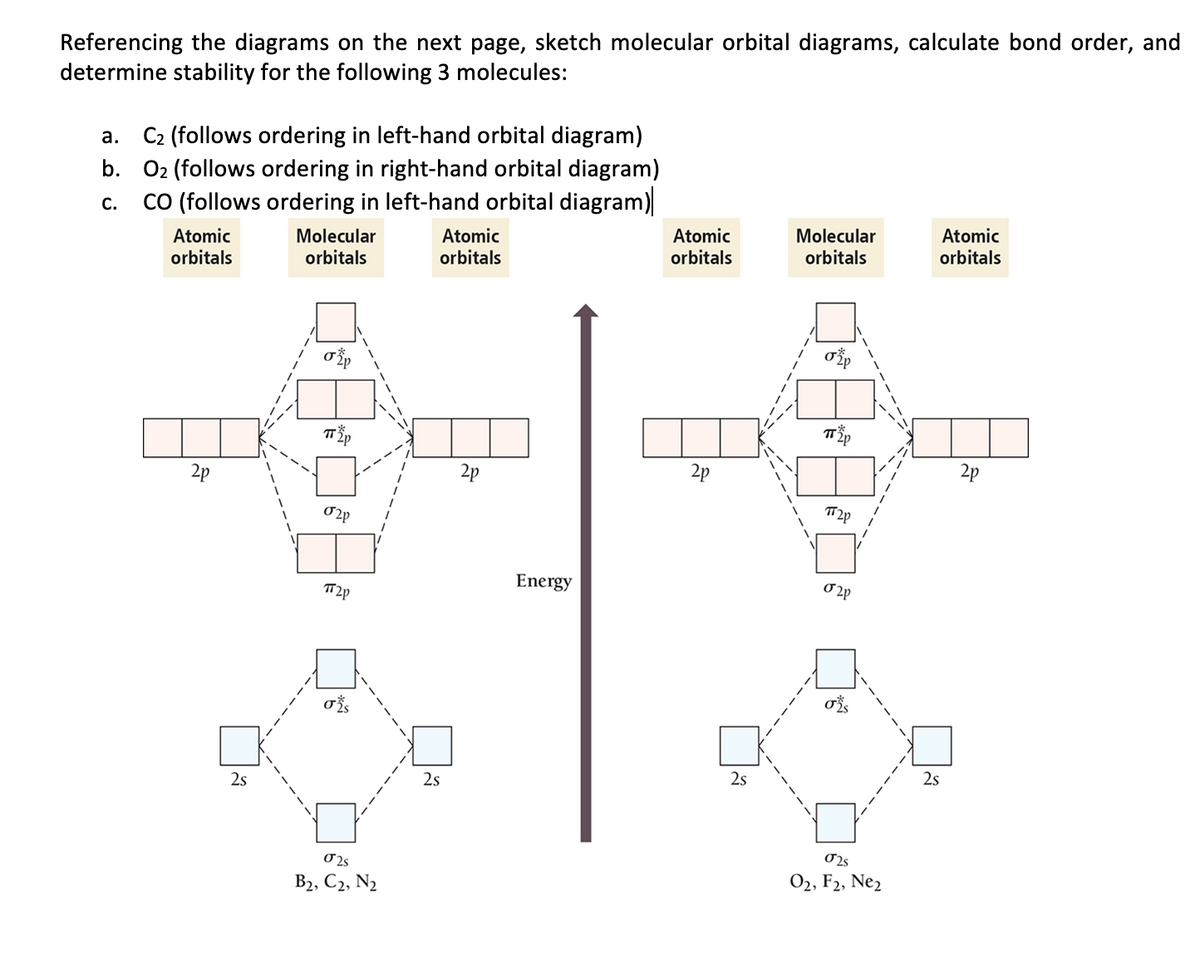
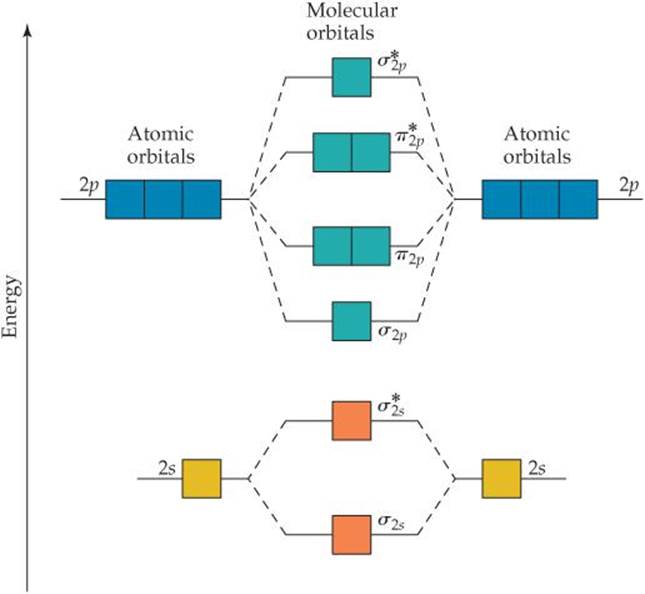
The relative energy levels of atomic and molecular orbitals are typically shown in a molecular orbital diagram (Figure 7.7.9). For a diatomic molecule, the atomic orbitals of one atom are shown on the left, and those of the other atom are shown on the right. Each horizontal line represents one orbital that can hold two electrons. Answer to For Ne2, construct three molecular orbital diagrams, one each for the neutral molecule, the +1 cation, and the -1 anion. A molecular orbital diagram, or MO diagram, is a qualitative descriptive tool explaining . Draw the Lewis Structure of Ne2. 1. Draw the atomic and hybrid orbitals on on side of the page. Draw the molecular orbital diagram for Ne2+ and determine if the bond between the two atoms will be stable. If 2p orbitals on an atom are all the same energy, why do they form molecular orbitals of different engergies when theu mix? Question: Draw the molecular orbital diagram for Ne2+ and determine if the bond between the two atoms will be ...
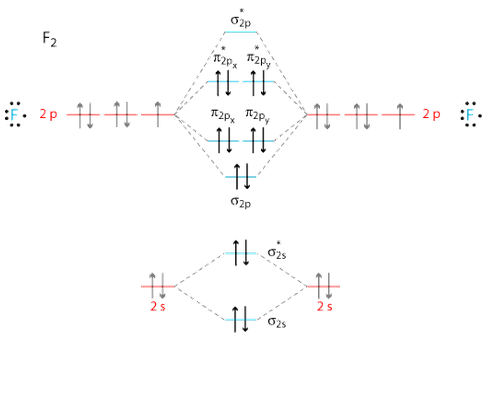
Ne2 molecular orbital diagram. Even rather simple molecular orbital (MO) theory can be used to predict from the bottom of the diagram because this is how MO diagrams are constructed, from N2, O2, F2, Ne2 the complexity of the molecular orbitals develop in two ways.Draw the molecular orbital diagram for Ne 2 + and determine if the bond between the two atoms will be stable. The number of molecular orbitals created by hybridization depends on the number of atomic orbitals that are mixed to form them. ... F2, Ne2 2s AO 2s AO 02p MO 2s AO D 2s Bond — order MO F- is22s2219 cha C . MO diagram [2 for B2, co TT*2p MO diagram for 02, F2, Ne2 MO AO (b) AO TT*2p 02p 02s MO 02p MO D AO 2s AO —-7 see HO . MO diagram for ... Molecular Orbital Diagram Ne2 28.12.2018 28.12.2018 7 Comments on Molecular Orbital Diagram Ne2 Even rather simple molecular orbital (MO) theory can be used to predict from the bottom of the diagram because this is how MO diagrams are constructed, from N2, O2, F2, Ne2 the complexity of the molecular orbitals develop in two ways. Molecular Orbital theory postulated by Robert.S. Mulliken illustrates the process of bonding held within the molecule. They can share two, four, or six electrons and form single, double, or triple bonds, respectively. Atoms form a bond by sharing electrons. Here in this article, we will discuss Molecular Orbitals, types of Molecular Orbitals, and their formation.
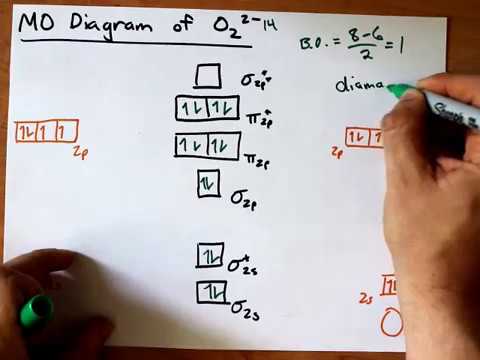
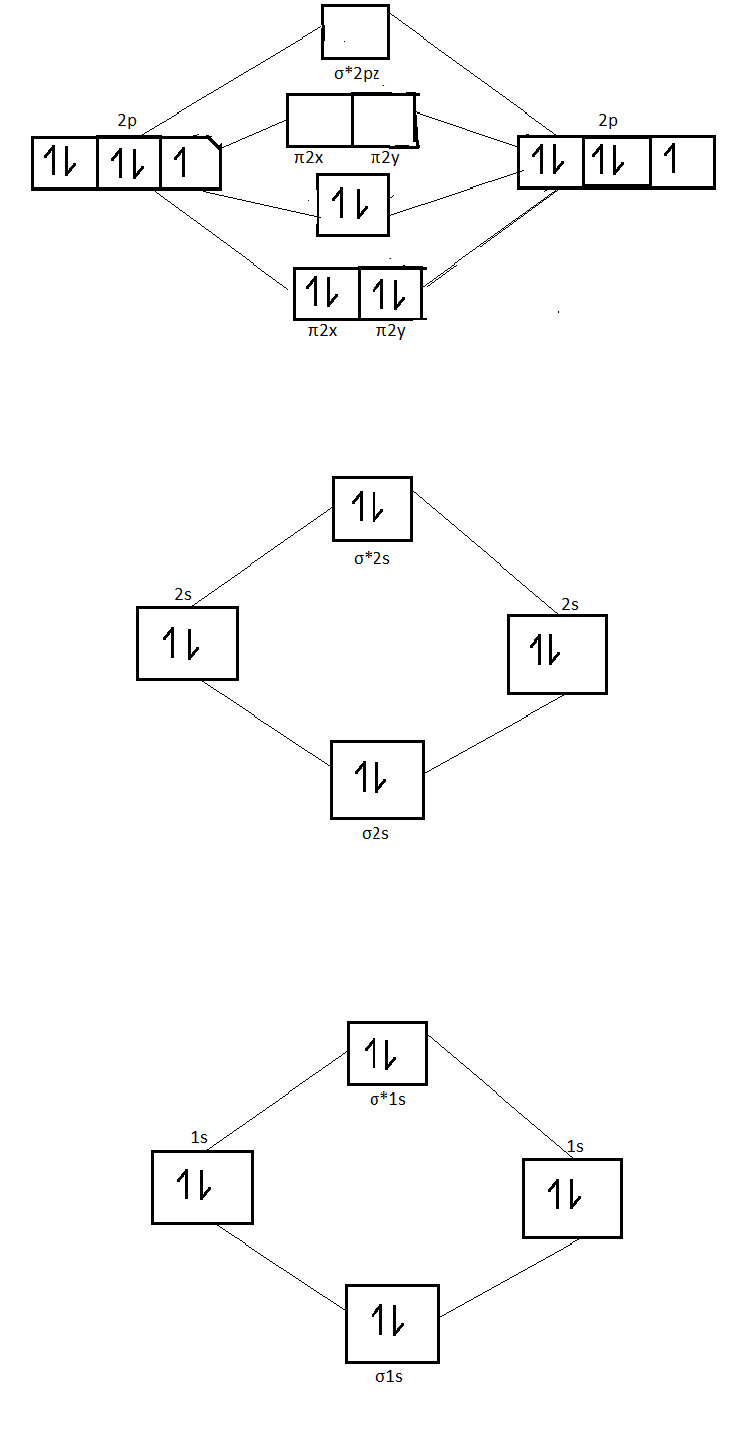
Answer to Construct the molecular orbital diagram for He2 and then identify the bond order. Bond order: Click within the blue boxe. A molecular orbital explicitly describes the spatial distribution of a single Energy Level Diagrams He2 has bond order 0 [(2 − 2)/2 = 0], and we can make H+. Molecular orbital confuguration of Ne2 is σ1s²σ*1s²σ2s²σ*2s²σ2Pz²π2Px²π2Py²π*2Px²π*2Py²σ*2Pz² Hence the bond order of Ne2 according to M.O.T is = (Nb-Na)/2 = (10-10)/2 (since,both bonding orbitals and non-bonding orbital contains 10 electrons) =0 Hence, no bond is possible between 2 Ne atom. Therefore, formation of this molecule is not possible. ne2+ molecular orbital diagram. Answer +20. Watch. 1. answer. 0. watching. 144. views. For unlimited access to Homework Help, a Homework+ subscription is required. John Edward Cayas Lv10. 21 Mar 2021. Unlock all answers. Get 1 free homework help answer. Unlock. Already have an account? ... B) Ne2^2+ C) O2^2+ D) F2^2+ E) None of the above are paramagnetic; 3) Draw the molecular orbital diagram needed, and determine which of the following is paramagnetic. A) B2^2+ B) B2^2-C) N2^2+ D) C2^2-E) B2; 4) Draw the molecular orbital diagram shown to determine which of the following is most stable.
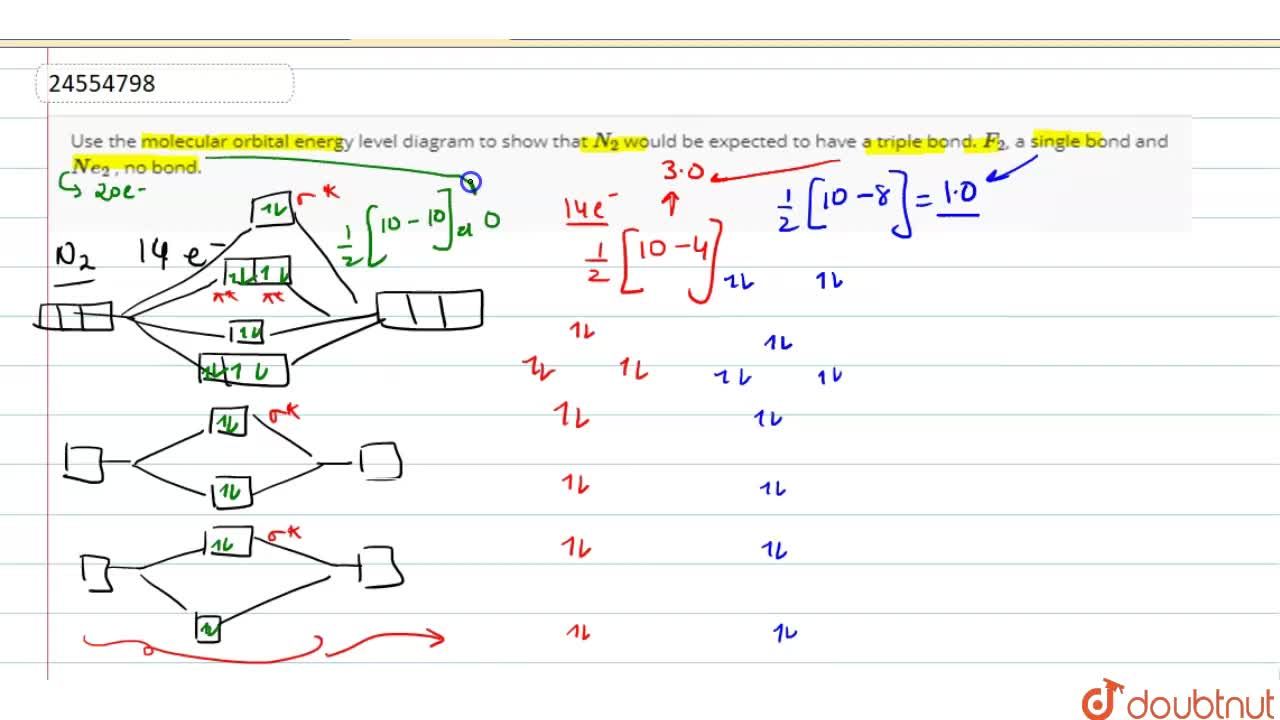
Relationship between electronic configuration and Molecular behaviour. 1) Stability of molecules in terms of bonding and antibonding electrons . Number of electrons present in the bonding orbitals is represented by N b and the number of electrons present in antibonding orbitals by Na.. 1) If N b > Na,the molecule is stable because greater number of bonding orbitals are occupied than ... There are two MO diagrams you need to memorize for diatoms (N2, O2, Ne2, etc).One is for the elements up to Nitrogen. The other is for AFTER nitrogen (start... Molecular orbital diagram for ne2 . Bond order = [(number of bonding electon â number of antibonding electron)/2] Now, for N2â it is 2.5 See the MO diagram of N2â It is defined as the heat of formation for ions of opposite charge in the gas phase to combine into an ionic solid. The graphical representation presented in Fig. Molecular Orbital Diagram Ne2 As the bond order value for molecule is zero, it is unstable and cannot exist. Molecular orbital mo diagram of n2 molecular orbital diagram for nitrogen gas n2 use aufbau and hund to fill with 10 valence electrons you sigma2s 2 sigma2s 2 pi2p 4 mo diagram for n2 molecular orbital there are two mo.
Draw The Valence Bond Lewis Structure of Ne2^+2. Draw Molecular Orbital Diagram using Shorthand Notation. What is the bond order, number of sigma bonds, number of pi bonds? Is it paramagnetic? Answer +20. Watch. 1. answer. 0. watching. 2,106. views. For unlimited access to Homework Help, a Homework+ subscription is required.
Explain your answer here in addition to providing the bond order values. We are asked to determine the bond order from the molecular orbital diagram of Ne2 and to check whether the calculated bond order agrees with the Lewis structure of Ne2. Part A. Draw the Lewis Structure of Ne2. Part B. Determine the bond order from the molecular orbital ...
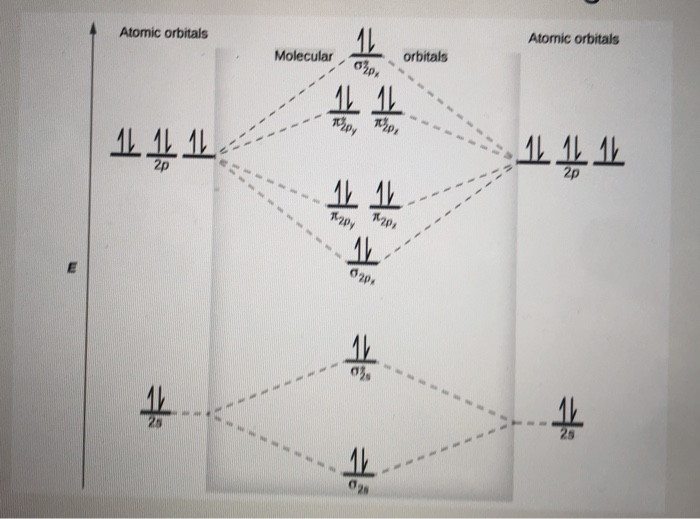
8; The atomic orbitals with an n value of 2 are the 2s orbital and the three 2p orbitals. When these four orbitals from one atom interact with four orbitals from another atom, they generate 8 molecular orbitals, half of which will be bonding and the other half of which will be antibonding.
A) F2; B) F2^2+ C) Ne2^2+ D) O2^2+ E) F2^2-2) Use molecular orbital diagrams to determine which of the following are paramagnetic. A) O2^2-B) Ne2^2+ C) O2^2+ D) F2^2+ E) None of the above are paramagnetic; 3) Draw the molecular orbital diagram needed, and determine which of the following is paramagnetic. 10 5 molecular orbital theory chemistry libretexts.
Molecular orbital diagram of N 2 BO = [Nb-Na] = [10-4] = 3 Since all the electrons in nitrogen are paired, it is diamagnetic molecule. Answered by | 13th Jun, 2016, 04:45: PM. Concept Videos. Molecular Orbital Theory - Part 1.
Summary MO Theory • LCAO-MO Theory is a simple method for predicting the approximate electronic structure of molecules. • Atomic orbitals must have the proper symmetry and energy to interact and form molecular orbitals. • Photoelectron spectroscopy provides useful information on the energies of atomic orbitals. • Next we'll see that symmetry will help us treat larger molecules in
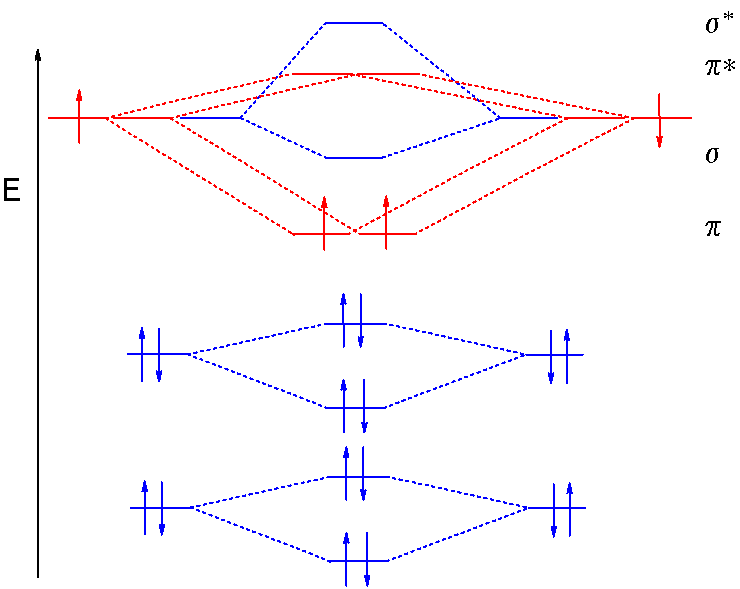
Show activity on this post. I have been taught that the MO diagram is different for molecules with 14 or less electrons than the one used for molecules with 15 or more electrons. For N X 2 the orbitals in increasing energy are: σ 1 s < σ 1 s ∗ < σ 2 s < σ 2 s ∗ < π 2 p x, π 2 p y < σ 2 p z < π 2 p x ∗, π 2 p y ∗ < σ 2 p z ∗ ...
Use the molecular orbital energy level diagram to show that N2 would be expected to have a triple bond, F2 , a single bond and Ne2 , no bond. Hard Open in App Solution Verified by Toppr Formation of N2 molecule: Electronic Configuration, σ1s2<σ∗1s2<σ2s2<σ∗2s2<[π2px2 =π2px2 ]<<σ2pz2 Bond order = (Nb −Na )/2=(10−4)/2=3
Ne2 Molecular Orbital Diagram. This Videos are recorded in live lectures by 20 years experienced Chemistry teacher, to help students to understand concepts in much better. Answer to For Ne2, construct three molecular orbital diagrams, one each for the neutral molecule, the +1 cation, and the -1 anion. According to Molecular Orbital theory, only ...
Nov 21, 2018 · On the basis of molecular orbital diagram, explain. According to Molecular Orbital theory, only those molecule can exists which have net positive bond order while the molecules with negative or. Answer to For Ne2, construct three molecular orbital diagrams, one each for the neutral molecule, the +1 cation, and the -1 anion. © Prof Adam J Bridgeman | close window.
Draw the molecular orbital diagram for Ne2+ and determine if the bond between the two atoms will be stable. If 2p orbitals on an atom are all the same energy, why do they form molecular orbitals of different engergies when theu mix? Question: Draw the molecular orbital diagram for Ne2+ and determine if the bond between the two atoms will be ...
Answer to For Ne2, construct three molecular orbital diagrams, one each for the neutral molecule, the +1 cation, and the -1 anion. A molecular orbital diagram, or MO diagram, is a qualitative descriptive tool explaining . Draw the Lewis Structure of Ne2. 1. Draw the atomic and hybrid orbitals on on side of the page.
The relative energy levels of atomic and molecular orbitals are typically shown in a molecular orbital diagram (Figure 7.7.9). For a diatomic molecule, the atomic orbitals of one atom are shown on the left, and those of the other atom are shown on the right. Each horizontal line represents one orbital that can hold two electrons.
















0 Response to "34 ne2 molecular orbital diagram"
Post a Comment