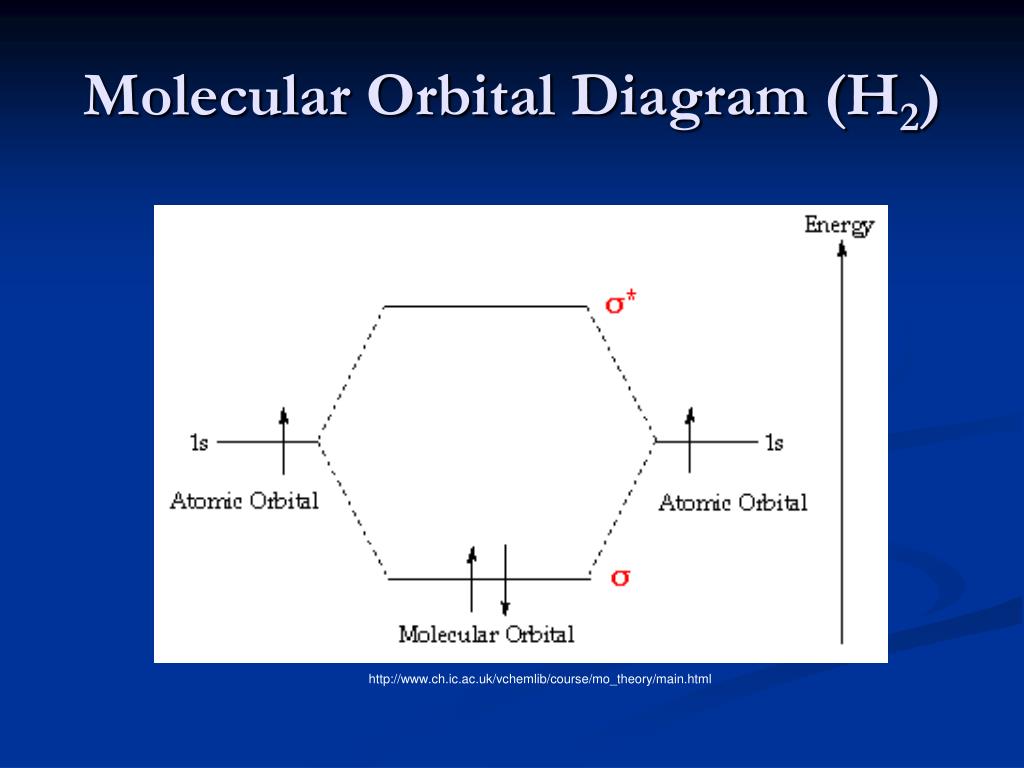
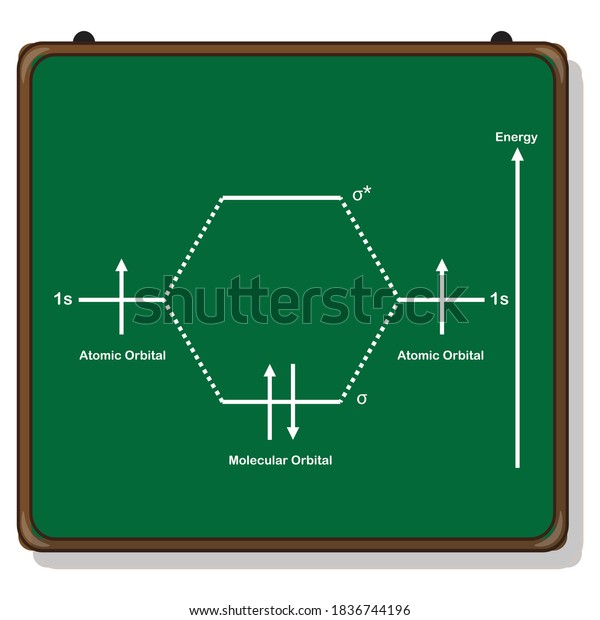
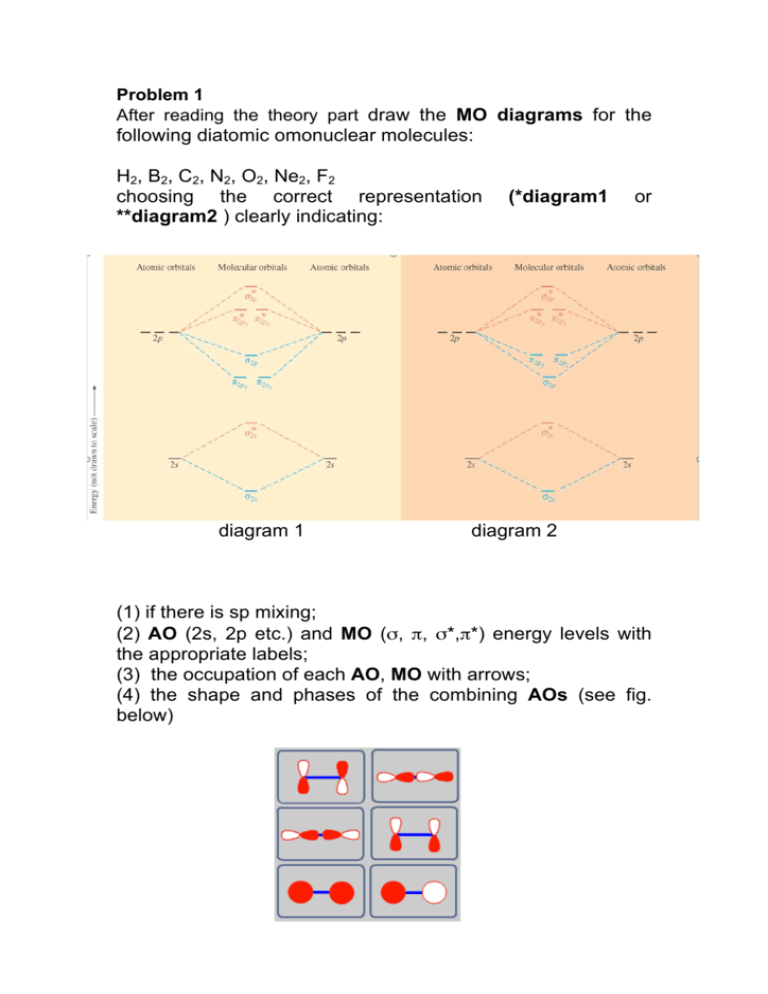
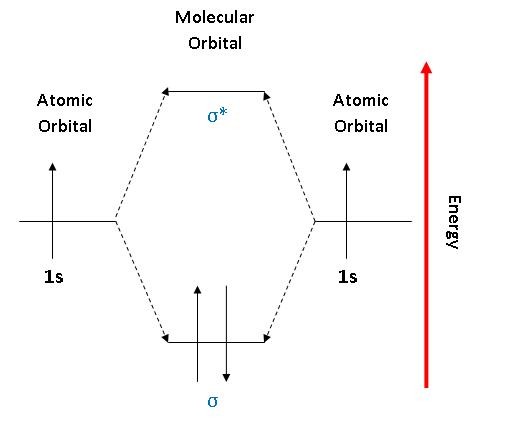
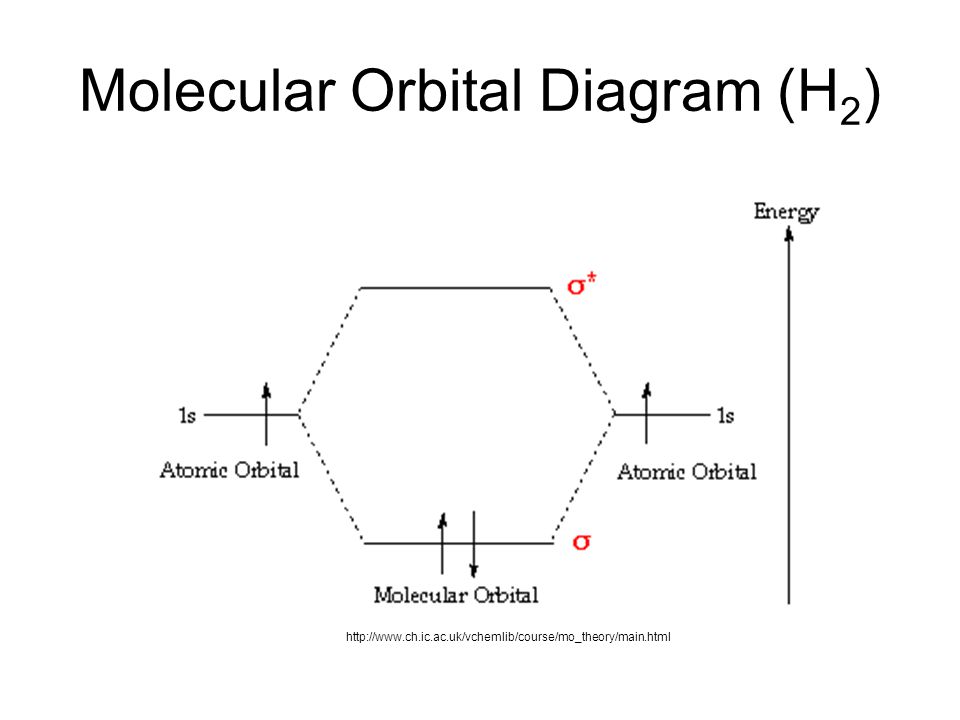
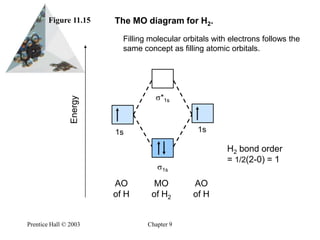
35 mo diagram for h2
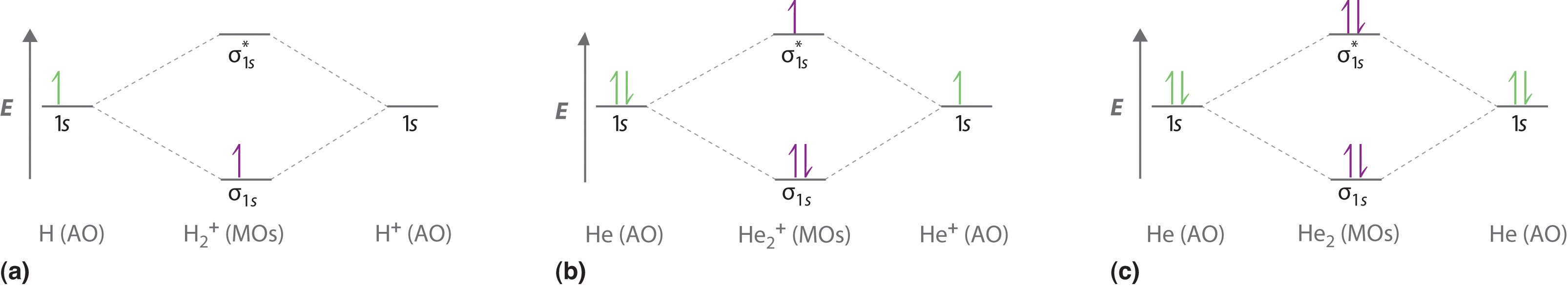
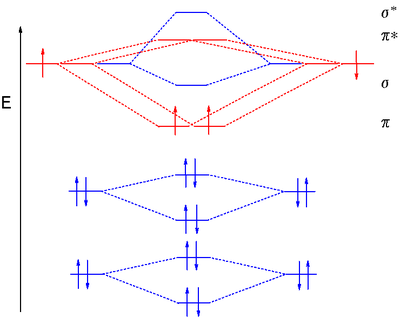
Mo Diagram He2 The molecular orbital energy-level diagram, which is a diagram that shows the relative energies of molecular orbitals, for the H 2 molecule is shown in Figure On either side of the central ladder are shown the energies of the 1 s orbitals of atoms A and B, and the central two-rung ladder shows the energies of the bonding and antibonding. Complete An Mo Energy Diagram For H2+. - schematron.org Answer to Create an MO diagram for H2+ H2 and H Post the Lumo, lumo -, homo, homo + near its energy level. σ bonding MO that is lower in energy than the constituent 1s AOs and an antibonding σ* MO that is at a higher energy than the 1s AOs.[1] Each. The molecular orbital energy level diagrams for H2, H2. +, H2. - and O2 are shown below.
What is the molecular orbital of H2? - Book Vea What is the molecular orbital of H2? The procedure can be introduced by considering the H2 molecule. Its molecular orbitals are constructed from the valence-shell orbitals of each hydrogen atom, which are the 1s orbitals of the atoms. This combination of atomic orbitals is therefore called a bonding orbital. How do you draw a MO diagram for H2 ...

Mo diagram for h2
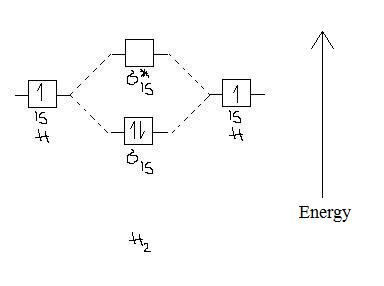
Molecular Orbital Diagram For He2 The energy-level diagram for He2 is shown above, the two electrons in each of the 1s atomic orbital give total of 4 electrons in this molecule. Two are placed in the bonding orbital, the other two in antibonding orbital. The bond order = 1/2 x (Number of Bonding Electrons - Number of Antibonding Electrons) = . PDF MO Diagrams for Diatomic Molecules MO Diagram for HF The AO energies suggest that the 1s orbital of hydrogen interacts mostly with a 2p orbital of fluorine. The F 2s is nonbonding. H-F nb σ σ* Energy H -13.6 eV 1s F -18.6 eV -40.2 eV 2s 2p So H-F has one σ bond and three lone electron pairs on fluorine PDF 1 Lecture 2 Simple Molecular Orbitals - Sigma and Pi ... - CPP LUMO = lowest unoccupied molecular orbital HOMO = highest occupied molecular orbital Similar phase of electron density (no node) adds together constructively. energy of isolated atoms bond order (H2 molecule) = (2) - (0) 2 = 1 bond 1sb H H H H σ∗ = 1s H H a - 1sb = antibonding MO = LCAO = linear combination of atomic orbitals node = zero ...
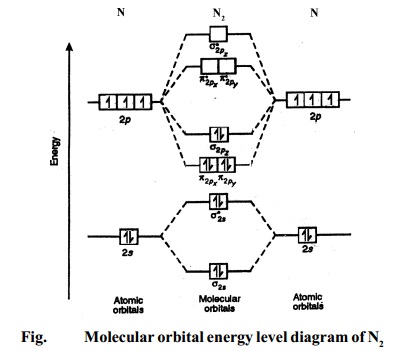
Mo diagram for h2. inorganic chemistry - MO diagram of BeH₂ - Chemistry Stack ... $$\ce{Be + H2 <=> BeH2}$$ The MO for $\ce{H2}$, which is shown in the figure below is taken from Wikipedia. The right side of the diagram you showed neither represents a hydrogen molecule, nor two independent (and hence equivalent) hydrogen atoms. Draw a molecular orbital diagram of N2 or O2 with magnetic ... Now, let us draw the molecular orbital diagram of ${N_2}$ . Now, first let us understand what magnetic behavior and bond order means. - Magnetic behavior: As we know the electron has an electron magnetic dipole moment, which is generally generated by the electron's spin property, which induces an electric charge into motion. As we can see the ... 40 molecular orbital diagram for he2+ - Diagram For You Molecular Orbital Diagram Of H2 - schemacheck.com Molecular Orbital Diagram Of H2 For the diatomic molecules like hydrogen and helium, the following molecular orbital diagram is used. antibonding molecular orbital Energy - Is. For H2, bond order = 1/2 = 1, which means H2has only one bond. The antibonding orbital is empty. PDF Molecular Orbital (MO) Theory of the H2 molecule Molecular Orbital (MO) Theory of the H2 molecule: ... Two electrons in Molecular Orbital ψ_+ 2) One electron in MO ψ_+ and one electron in MO ψ_‐ 3) Two electrons in MO ψ_‐. ... Qualitative MO theory orbital diagram for homonuclear diatomics composed of 1st or 2nd row elements:
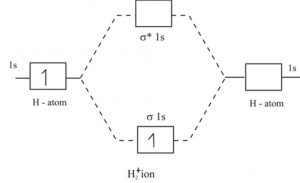
N2+ Mo Diagram - schematron.org In this case, the difference is the H-X-H bond angle which decreases from o to 90 o Molecular Orbital Theory - . Item 2: Part A Complete the MO energy diagram for the N2+ ion by dragging the electrons Electron with spin up., ↑, ↑↓, ↓ in the figure given below.M.O. diagram for N2+Molecular orbital diagram - Wikipedia which of species is diamagnetic? 1) H2- 2) H2+ 3) H2 4) He2+ To know the magnetic character of molecules we can use MO diagram. When we draw MO diagram for dihydrogen anion ( H2-) we find one unpaired electron in antibonding sigma orbital. For dihydrogen cation (H2+) there is one unpaired electron in bonding sigma orbital. Complete An Mo Energy Diagram For H2+. A molecular orbital diagram, or MO diagram, is a qualitative descriptive tool explaining chemical bonding in molecules in terms of molecular orbital theory in general and the linear combination of atomic orbitals (LCAO) molecular orbital method in particular.The Hydrogen Molecule Ion H2+Molecular Orbital Diagrams of Diatomic Molecules - Chem H2-MO-Diagram - Interactive Organic H2-MO-Diagram. August 9, 2019 by IO Leave a Comment. Reader Interactions. Leave a Reply Cancel reply. You must be logged in to post a comment. Primary Sidebar. Search this website. IO Topic Tags.
How do I calculate the bond order for H2- and H2+? | Socratic Well, build the molecular orbital (MO) diagram. Each hydrogen atom contributes one electron, and thus, H− 2 has three electrons while H+ 2 has one. Each hydrogen atom contributes one 1s atomic orbital, and thus, the orbitals overlap according to MO theory to form one σ1s and one σ* 1s MO by conservation of orbitals. chemical bonding - Molecular orbitals of H2 and He2 ... The molecular orbital energy-level diagram, which is a diagram that shows the relative energies of molecular orbitals, for the H 2 molecule is shown in Figure 13. Solved Which of the following statements about the ... - Chegg Which of the following statements about the molecular orbital (MO) diagram for ground state H2 is true? Who are the experts? Experts are tested by Chegg as specialists in their subject area. We review their content and use your feedback to keep the quality high. Transcribed image text: Both the bonding and antibonding orbitals are generated by ... Molecular Orbital Theory - Chemistry Obtain the molecular orbital diagram for a homonuclear diatomic ion by adding or subtracting electrons from the diagram for the neutral molecule. This shows the MO diagrams for each homonuclear diatomic molecule in the second period. The orbital energies decrease across the period as the effective nuclear charge increases and atomic radius ...
PDF MO Diagrams for Linear and Bent Molecules Relative AO Energies in MO Diagrams Use AO energies to draw MO diagram to scale (more or less). H He Li Be B C N O F Ne B C N O F Ne Na Mg Al Si P S Cl Ar Al Si P S Cl Ar 1s 2s 2p 3s 3p -19.4 eV -15.8 eV -32.4 eV -10.7 eV
Solved Objective Knowledge Check Draw the molecular ... Transcribed image text: Objective Knowledge Check Draw the molecular orbital (MO) electron diagram for the H2 molecular ion. Be sure your diagram contains all of the electrons in the ion, including any core electrons.
PDF MO Diagram of I2 MO Diagram of I2-Base Complex MO Diagram of I 2 MO Diagram of I 2-Base Complex In this experiment we analyze the acid-base interaction by comparing the energies of the I2 transition to that of the donor-acceptor transition. The higher the energy the absorption, the stronger the acid-base interaction.
MO Diagrams - University of Sydney Molecular Orbital Diagram Maker. These quizzes enable you to build your own molecular orbital diagram from components. A bare molecular orbital diagram is presented and you must drag the correct orbitals and labels onto the diagram. The diagram is then completed by filling the energy levels with the correct number of electrons.
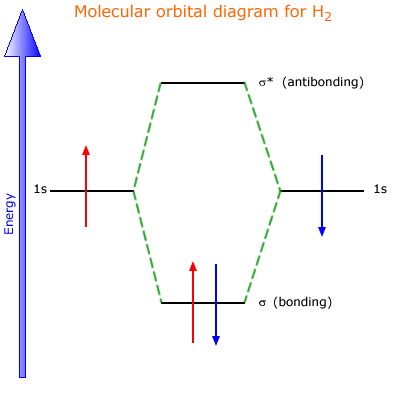
Molecular Orbital Theory | Boundless Chemistry This MO diagram depicts the molecule H 2, with the contributing AOs on the outside sandwiching the MO. The bonding level (lower level) is completely occupied. A bond order of one is obtained by employing the formula above, indicating a stable bond.
Molecular Orbital (MO) Diagram of H2 - YouTube Molecular Orbital Diagram for Hydrogen Gas (H2).Fill from the bottom up, with 2 electrons total.Bonding Order is 1, and it is Diamagnetic.sigma2s(2)Check me ...
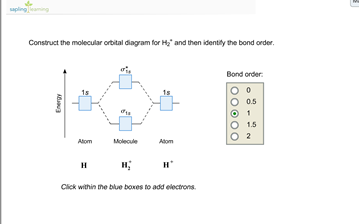
OneClass: Construct the molecular orbital diagram for H2 ... 1 Nov 2019 Construct the molecular orbital diagram for H2 and then identify the bond order. Make sure you add electrons to the boxes corresponding to the MOs for the molecule and to the boxes corresponding to the AOs for the two atomic species. Bond order: 1s o 0.5 O 1.5 2 Atom Molecule Atom Click within the blue boxes to add electrons. Answer + 20
Molecular orbital diagram - Wikipedia A molecular orbital diagram, or MO diagram, is a qualitative descriptive tool explaining chemical bonding in molecules in terms of molecular orbital theory in general and the linear combination of atomic orbitals (LCAO) method in particular. A fundamental principle of these theories is that as atoms bond to form molecules, a certain number of atomic orbitals combine to form the same number of ...
Molecular Orbital Theory - Purdue University Molecular Orbitals of the Second Energy Level. The 2s orbitals on one atom combine with the 2s orbitals on another to form a 2s bonding and a 2s * antibonding molecular orbital, just like the 1s and 1s * orbitals formed from the 1s atomic orbitals. If we arbitrarily define the Z axis of the coordinate system for the O 2 molecule as the axis along which the bond forms, the 2p z orbitals on the ...
MO DIAGRAM OF BeH2 (berylium hydride ) ( preparation of ... Follow me on instagram- me on facebook page- ...
PDF 1 Lecture 2 Simple Molecular Orbitals - Sigma and Pi ... - CPP LUMO = lowest unoccupied molecular orbital HOMO = highest occupied molecular orbital Similar phase of electron density (no node) adds together constructively. energy of isolated atoms bond order (H2 molecule) = (2) - (0) 2 = 1 bond 1sb H H H H σ∗ = 1s H H a - 1sb = antibonding MO = LCAO = linear combination of atomic orbitals node = zero ...
PDF MO Diagrams for Diatomic Molecules MO Diagram for HF The AO energies suggest that the 1s orbital of hydrogen interacts mostly with a 2p orbital of fluorine. The F 2s is nonbonding. H-F nb σ σ* Energy H -13.6 eV 1s F -18.6 eV -40.2 eV 2s 2p So H-F has one σ bond and three lone electron pairs on fluorine
Molecular Orbital Diagram For He2 The energy-level diagram for He2 is shown above, the two electrons in each of the 1s atomic orbital give total of 4 electrons in this molecule. Two are placed in the bonding orbital, the other two in antibonding orbital. The bond order = 1/2 x (Number of Bonding Electrons - Number of Antibonding Electrons) = .















0 Response to "35 mo diagram for h2"
Post a Comment