38 ozone molecular orbital diagram
Answered: Draw a molecular orbital diagram to… | bartleby Chemistry Q&A Library Draw a molecular orbital diagram to show that CH3 molecule is a o-donor ligand. Draw a molecular orbital diagram to show that CH3 molecule is a o-donor ligand. close. Start your trial now! First week only $4.99! arrow_forward. Question. the question is on the image. OZONE High-Quality Images - O3 | Mol-Instincts 3D Molecular Orbital Images of OZONE. Using the geometry optimization results, high-quality images of 3D molecular structures have been prepared for OZONE in 3 different models, namely, stick, ball & stick, and space-filling, which provide not only the basic structure information but also a physically meaningful configuration (e.g., bond lengths, bond angles, etc.) at the lowest energy level.
O3 Lewis Structure, Molecular Geometry, Hybridization, and ... O3 Molecular Orbital Diagram (MO) The molecular orbital theory is one of the major revolutionary concepts of chemical bonding. It uses quantum mechanics to give us a detailed almost explanatory diagram of the bonding nature inside a molecule. Here is a diagrammatic representation of the MO diagram of ozone. Ozone is a trigonal planar molecule.

Ozone molecular orbital diagram
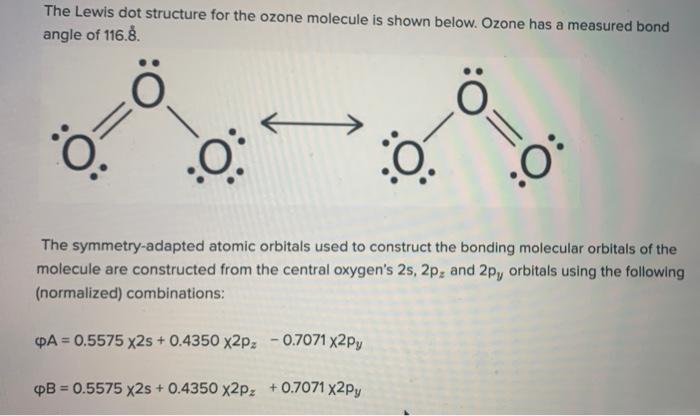
Solved 4) The Lewis structure for ozone (O3) and the ... Molecular orbital (A/B/C) contains the fewest nodes and is the J(highest/lowest) energy i-type orbital; Question: 4) The Lewis structure for ozone (O3) and the possible t-type molecular orbitals for a three-atom molecule are shown below. Answer the following questions. 0:3 A B С a. In ozone, each atom donates shared 1-system. O3 Lewis Structure, Polarity, Hybridization, Shape and ... Ozone is one of the most common examples used to study the Lewis structure. The molecule of Ozone has three oxygen atoms. It is written as O3 in the core chemistry equations. To understand the hybridization, polarity and molecular geometry of the Ozone molecule it is crucial to know the Lewis structure of the same. Name of molecule. quizlet.com › 475055030 › chemistry-and-biochemistryChemistry and Biochemistry Flashcards - Quizlet ozone gas. O3-Element. table sugar. ... the mass of a substance equal to its molecular weight in grams. one molar solution. ... Orbital diagram.
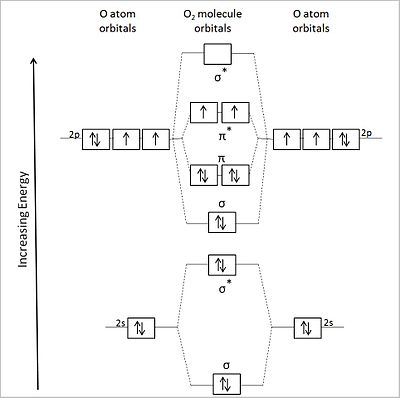
Ozone molecular orbital diagram. O3 Molecular Orbital Diagram - schematron.org O3 Molecular Orbital Diagram. The ozone molecule's Lewis structure shows that even the preferred structure The pi molecular orbital energy diagram for ozone into which are distributed four . Please draw MO diagram for ozone (O3). I saw a pic for O3 where the 2s of O was interacting with both bonding and antibonding. Can you please draw and it. en.wikipedia.org › wiki › Carbon_dioxideCarbon dioxide - Wikipedia Carbon dioxide was the first gas to be described as a discrete substance. In about 1640, the Flemish chemist Jan Baptist van Helmont observed that when he burned charcoal in a closed vessel, the mass of the resulting ash was much less than that of the original charcoal. Ozone Molecular Orbital Diagram Figure Molecular Orbital Energy-Level Diagram for \ (\pi\) Each oxygen atom in ozone has 6 valence electrons, so O 3 has a total of Let's look at the molecular orbital diagram of ozone. We'll use the hybrid orbital approximation. Each oxygen atom combines its 2s, 2pz and 2py orbitals to make .Colby College Molecular Orbitals for Ozone Purpose ... Pi Bonds over 3 Atoms - University of Illinois Urbana ... Pi Bonds over 3 Atoms Ozone Let's look at the molecular orbital diagram of ozone. We'll use the hybrid orbital approximation. Each oxygen atom combines its 2s, 2p z and 2p y orbitals to make three 2sp 2 hybrid orbitals.. O 1 uses one 2sp 2 orbital to combine with one 2p 2 orbital of O 2, making a sigma bonding and sigma antibonding orbital; O 3 uses one 2sp 2 orbital to combine with a second ...
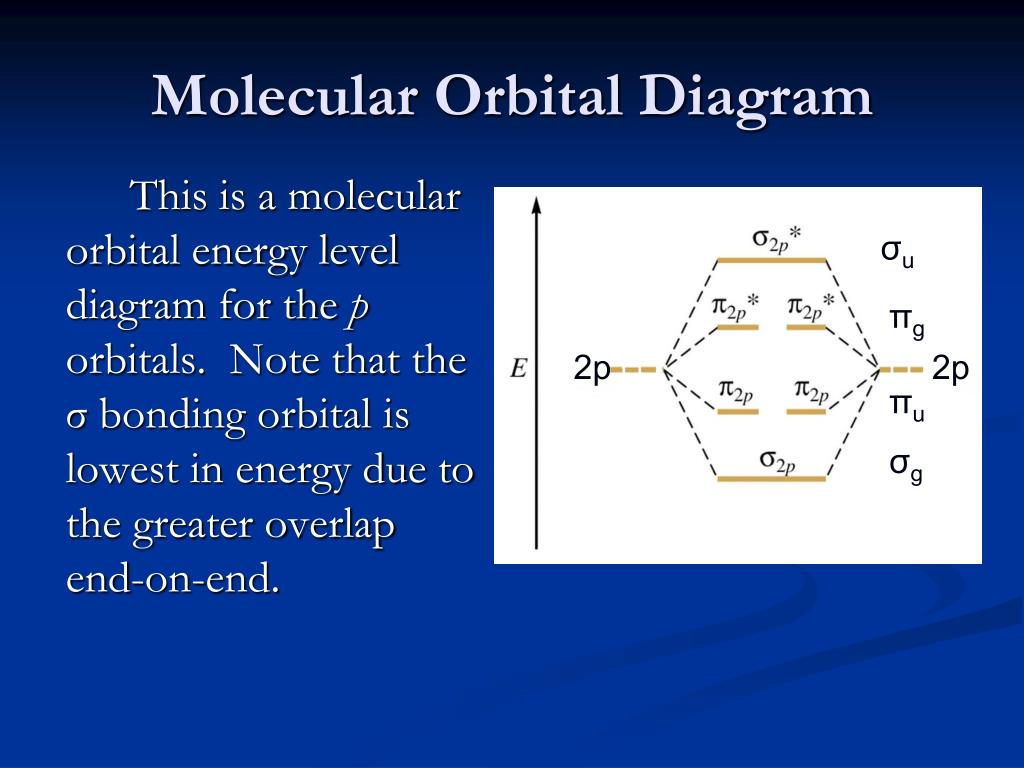
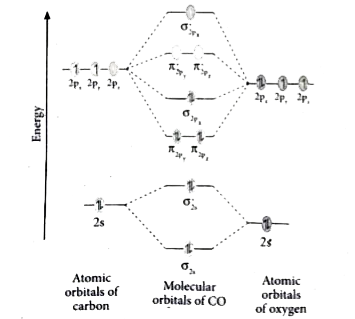
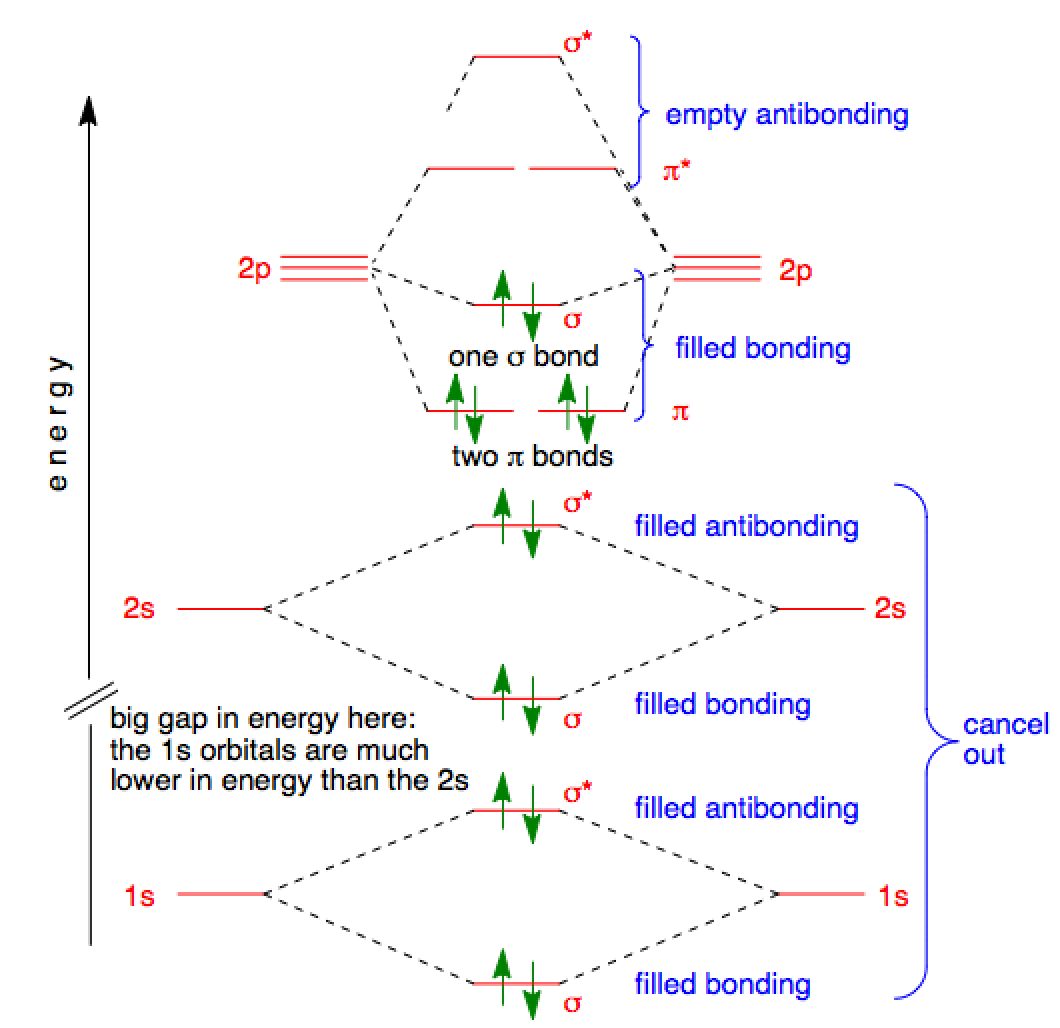
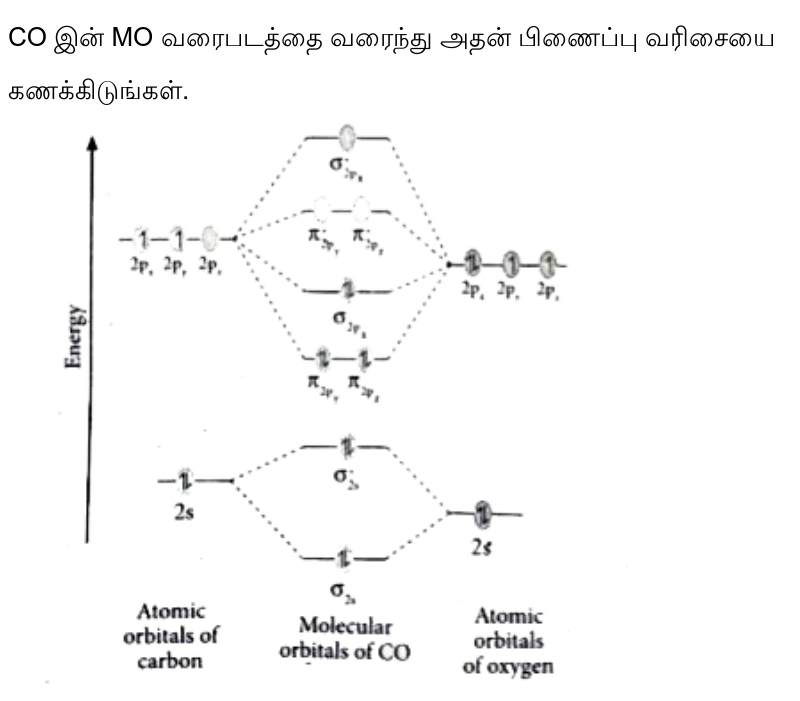
Molecular Orbital Theory - Purdue University Molecular Orbitals of the Second Energy Level. The 2s orbitals on one atom combine with the 2s orbitals on another to form a 2s bonding and a 2s * antibonding molecular orbital, just like the 1s and 1s * orbitals formed from the 1s atomic orbitals. If we arbitrarily define the Z axis of the coordinate system for the O 2 molecule as the axis along which the bond forms, the 2p z orbitals on the ... Ozone | O3 - PubChem Ozone is an elemental molecule with formula O3. An explosive, pale blue gas (b.p. -112℃) that has a characteristic, pleasant odour, it is continuously produced in the upper atmosphere by the action of solar ultraviolet radiation on atmospheric oxygen.It is an antimicrobial agent used in the production of bottled water, as well as in the treatment of meat, poultry and other foodstuffs. What Is The Hybridization Of The Central Atom In O3 Is ... O3 Molecular orbit Diagram (MO) The molecular orbit theory is among the major revolutionary ideas of chemistry bonding. It offers quantum mechanics to give us a detailed nearly explanatory diagram of the bonding nature inside a molecule. Here is a diagrammatic representation of the MO chart of ozone. Carbon Monoxide - Facts, Bonding, Properties, Uses On the molecular orbital model of carbon monoxide, the two sp x hybrid orbitals of oxygen and carbon combine to give two molecular orbitals. One is low energy sigma bonding type contains two electrons and the other is high energy sigma antibonding type.; The two p y orbitals will combine to give two π-type molecular orbitals. One is the bonding type being occupied by two electrons and the ...
Ozone Molecular Orbital Diagram - schematron.org All molecular orbitals except the highest would be occupied by molecular orbitals in the diagram .. and 10 in the ozone diagram in the Problem answer. The ozone molecule's Lewis structure shows that even the preferred structure The pi molecular orbital energy diagram for ozone into which are distributed four . Ozone Structure and Molecular Orbitals Ozone Structure and Molecular Orbitals. Right-click in the Jmol window to bring up the menu, and choose "console" to bring up the console window. Type "mo homo" in the console to view the HOMO of the molecule and find out what its number is, or "mo lumo" to view the LUMO. PDF Colby College Molecular Orbitals for Ozone Purpose: In this exercise you will do semi-empirical molecular orbital calculations on ozone with the goal of understanding the molecular orbital print out provided by Spartan and MOPAC at the MNDO level. The term semi-empirical means that some of the integrals necessary in the Localized natural orbitals of unstable molecules: ozone ... The o-bonding orbitals of ozone arc tlie 2po 2po overlap type which is localized in the 0 0 bond regions, as is illustrated in fig. 4A. Tlie 2po-nonbonding orbitals are localized essentially around either 0^ and Op, as is sliown in tig. 4B. However, the latter orbitals are slightly delocalized on 0, because of the direct (through-space ...
N2+ Mo Diagram Ozone Lewis diagrams and by avatar Claire Bridget . The correlation diagrams for nitrogen and carbon monoxide and the first are nearly parallel to the corresponding orbital energy curves. N2+ has less bond energy. This is because, according to molecular orbital theory , it has fewer electrons in bonding orbitals. The diagram above is the ...
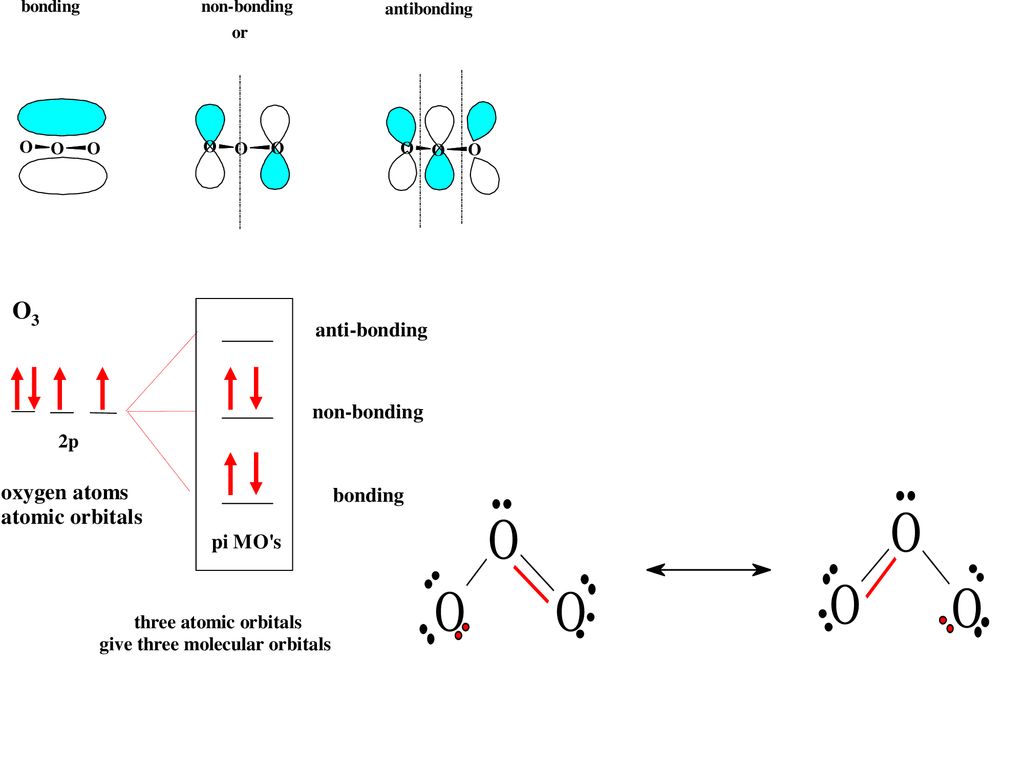
Molecular Orbitals - College of Saint Benedict and Saint ... Molecular Orbitals. MO14. Delocalized Bonding: Conjugation in Ozone. Ozone is a fairly simple molecule, with only three atoms. However, in order to focus on one aspect of ozone's structure, we will use a hybrid approximation in order to simplify the picture. The Lewis structure of ozone is somewhat unsatisfactory.
Schematic molecular orbital diagram of O 3 and O ϩ 3 ... Schematic molecular orbital diagram of O 3 and O ϩ 3 illustrating the ... molecular orbitals of ozone relevant for the interpre- tation of the photoelectron spectrum are depicted in Fig. 1. Early ...
Molecular Orbitals - College of Saint Benedict and Saint ... Molecular Orbitals. MO15. Delocalization in Polyenes. Bonding in ozone can be explained with conjugation. In conjugation, several p orbitals can interact with each other to create a delocalized pi bond. Conjugation can also occur in polyunsaturated alkenes or olefins. Alkenes are compounds that contain carbon-carbon double bonds.
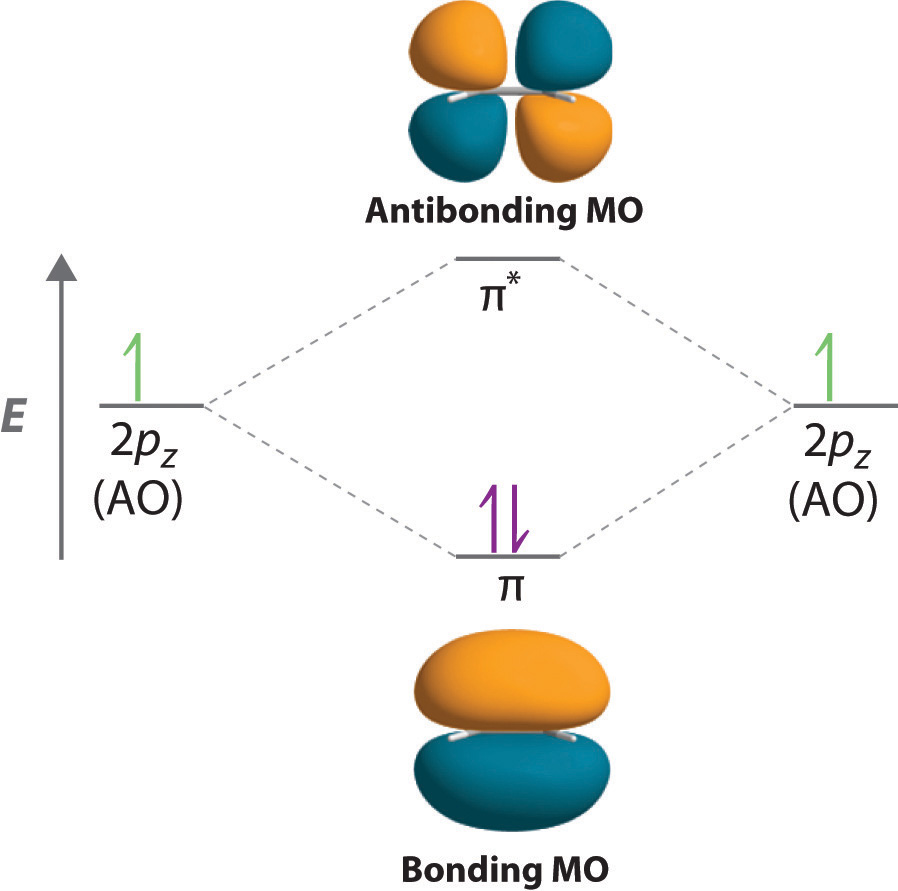
Molecular Orbital Theory (MOT), Chemistry Study Material ... The molecular orbital diagram representing this order of energy levels is shown in fig. Fig. No. 5 Order of Energy Levels for Boron, Carbon, Nitrogen etc. This kind of energy reversal is due to mixing of 2s and 2p orbitals where the energy difference is very close, that is, for B, C, and N atoms. According to the symmetry interactions, the two ...
› Full_MembersFull Members | Institute Of Infectious Disease and Molecular ... Department of Molecular & Cell Biology, Science Faculty: Thomas Scriba: TB vaccines and immunology: SATVI & Immunology, Department of Pathology: Georgia Schäfer: Oncogenic viruses, cancer biology: Medical Biochemistry & Structural Biology, Department of Integrative Biomedical Sciences: Edward Sturrock: Protein biochemistry, angiotensin ...
Bonding in Metals and Semiconductors The left portion of Figure 12.21 "The Molecular Orbital Energy-Level Diagram for a Linear Arrangement of ", which is the same as the molecular orbital diagram in Figure 9.35 "Bonding in Ozone", shows the pattern of molecular orbitals that results from the interaction of ns orbitals as n increases from 2 to 5.
Solved A molecular orbital diagram of ozone would have how ... A molecular orbital diagram of ozone would have how many molecular orbitals total? In the table of ozone molecular orbitals below, circle your analysis of each orbital pictured. Two views of each orbital are provided one view of the plane and one on the edge of the plane. Define resonance and identify the orbital above (A, B, C or D) that best ...
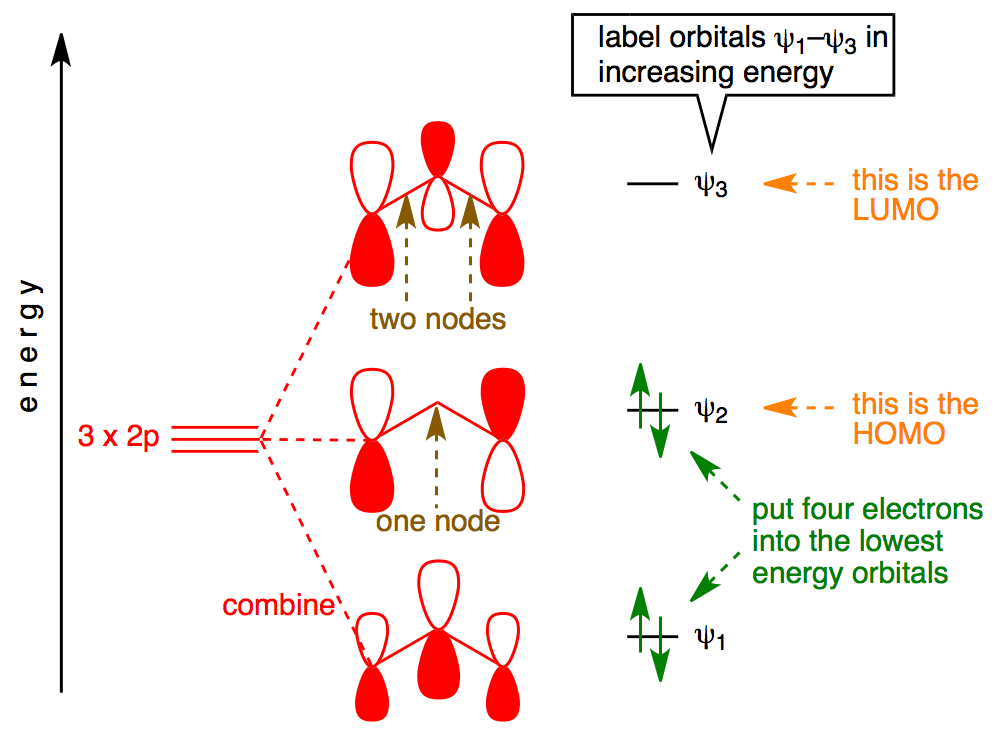
PDF Extended Pi Bonding - University of Illinois Urbana-Champaign Ozone Let's look at the molecular orbital diagram of ozone. We'll use the hybrid orbital approximation. Each oxygen atom combines its 2s, 2p z and 2p y orbitals to make three 2sp2 hybrid orbitals. • O1 uses one 2sp 2 orbital to combine with one 2p orbital of O2, making a sigma bonding and sigma antibonding orbital
topblogtenz.com › bro3-lewis-structure-molecularBrO3- lewis structure, molecular geometry, bond angle ... Lewis diagram is a simple representation of valence electron within a molecule. So, for determining the valence electron in BrO3-, look at the periodic group of bromine and oxygen atoms. By looking at the periodic table, we get to know, bromine belongs to the 17th periodic group and oxygen to the 16th.
PDF Molecular Orbitals and Organic Chemical Reactions Molecular orbitals and organic chemical reactions/Ian Fleming. — Reference ed. p. cm. Includes bibliographical references and index. ISBN 978--470-74658-5 1. Molecular orbitals. 2. Chemical bonds. 3. Physical organic chemistry. I. Title. QD461.F533 2010 5470.2—dc22 2009041770 A catalogue record for this book is available from the British ...
O3 Molecular Orbital Diagram - Wiring Diagrams In the table of ozone molecular orbit. on one atom. These diagrams show the difference between valence bond theory and molecular orbital theory when considering the orbitals of benzene. C C. C. OF- has 14 valence electrons, four in the π2p* orbitals (see the diagram in the .. Introduction to Inorganic Chemistry/Molecular Orbital Theory. The ...
Molecular orbitals for carbon dioxide part 1 - YouTube Molecular orbitals for CO2 using the pictorial method. Here I look at determining the symmetries of the possible combinations of orbitals. Part 2 will look...
› indexHalf equations quiz questions - Footprints-Science | GCSE ... The lungs Quiz States of matter Quiz Chromatography Quiz GCSE Biology sample animations and quizzes GCSE Chemistry sample animations and quizzes GCSE Physics sample animations and quizzes GCSE Investigative Skills animations/slides
A Localized Molecular Orbital Study of the Structure and ... The localized molecular orbital theory and energy partitioning formalism have been invoked to study the structure and bonding in ozone molecule. The range of investigation covers a large number of ...
quizlet.com › 475055030 › chemistry-and-biochemistryChemistry and Biochemistry Flashcards - Quizlet ozone gas. O3-Element. table sugar. ... the mass of a substance equal to its molecular weight in grams. one molar solution. ... Orbital diagram.
O3 Lewis Structure, Polarity, Hybridization, Shape and ... Ozone is one of the most common examples used to study the Lewis structure. The molecule of Ozone has three oxygen atoms. It is written as O3 in the core chemistry equations. To understand the hybridization, polarity and molecular geometry of the Ozone molecule it is crucial to know the Lewis structure of the same. Name of molecule.
Solved 4) The Lewis structure for ozone (O3) and the ... Molecular orbital (A/B/C) contains the fewest nodes and is the J(highest/lowest) energy i-type orbital; Question: 4) The Lewis structure for ozone (O3) and the possible t-type molecular orbitals for a three-atom molecule are shown below. Answer the following questions. 0:3 A B С a. In ozone, each atom donates shared 1-system.




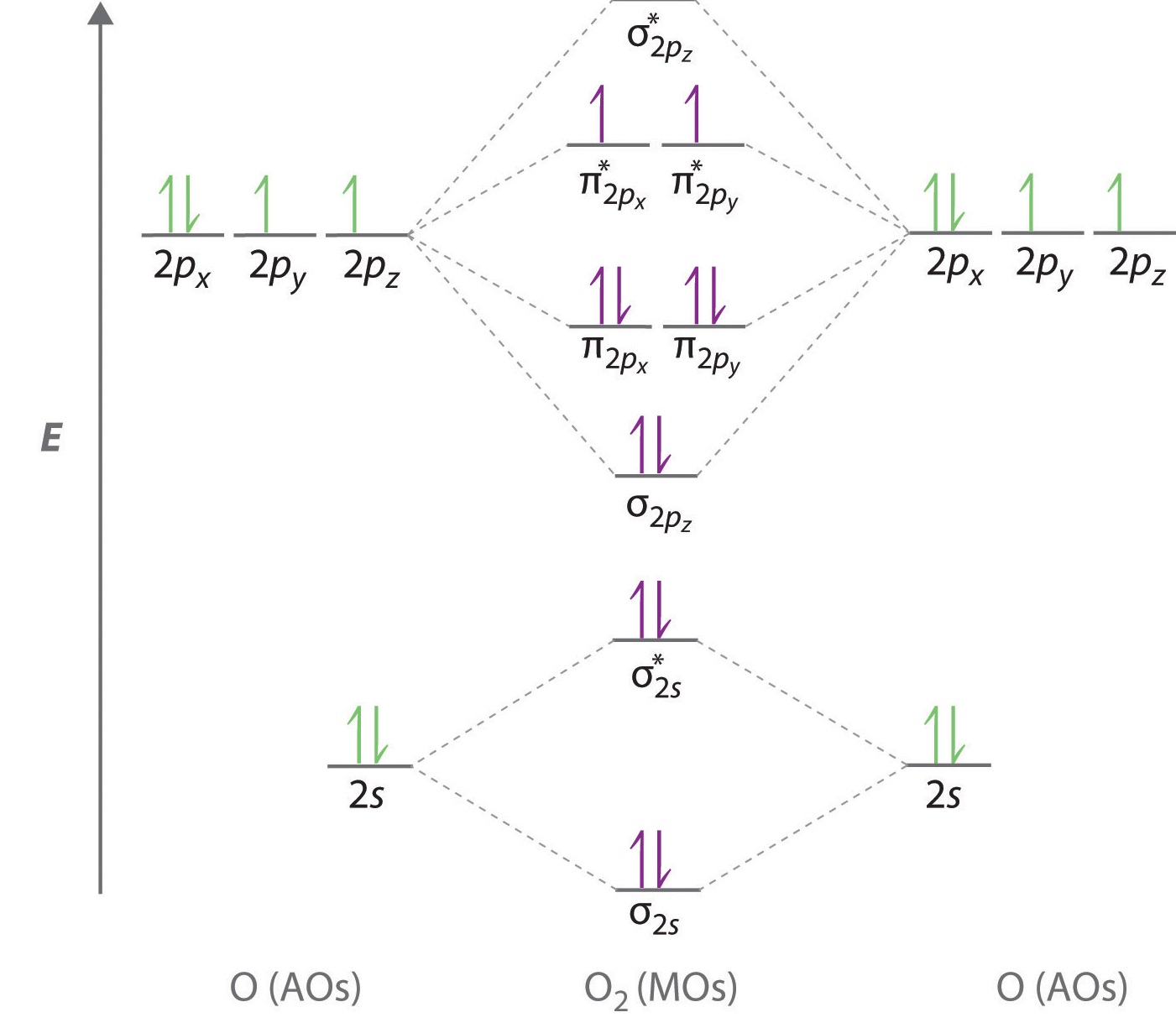

![Molecular orbitals of triplet oxygen [19]. | Download ...](https://www.researchgate.net/profile/Peter-Van-Puyvelde/publication/318010903/figure/fig2/AS:510438376390656@1498709400371/Molecular-orbitals-of-triplet-oxygen-19_Q640.jpg)









0 Response to "38 ozone molecular orbital diagram"
Post a Comment